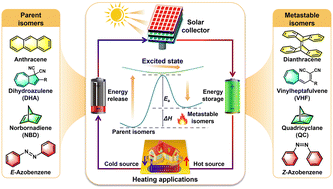
Introduction: The Heat Problem Nobody Talks About
Here's something that might surprise you: heating accounts for nearly half of global energy demand. Not electricity, not transportation, but heating. Keeping buildings warm, powering industrial processes, cooking food, providing hot water. Yet when we talk about the clean energy transition, we obsess over electricity and solar panels that generate it. We've gotten genuinely good at storing solar electricity in lithium-ion batteries. A decent battery pack can hold energy for hours or days without much loss. But heat? That's where the energy industry hits a wall.
The problem is asymmetrical. Electricity is easy to store. Heat is hard. You can't just stick heat in a battery and pull it back out six months later. Heat dissipates. It radiates away. It diffuses into the surroundings. And here's where it gets tricky: roughly two-thirds of the heat demand on the planet still comes from burning fossil fuels. Natural gas, oil, and coal work because they store vast amounts of energy in their chemical bonds—energy that accumulated over millions of years of sunlight hitting ancient organisms.
What if you could do that on purpose? What if you could take sunlight hitting your roof right now and trap it inside the bonds of a specially engineered molecule, then store that molecule in a tank in your basement? And then, months later when winter hits, you crack open those bonds and extract the heat on demand?
That's the promise of molecular solar thermal energy storage, or MOST. It's a category of technology that's been "the next big thing" for roughly thirty years. Real researchers have worked on it seriously. Real labs have funding. Real papers get published. Yet somehow it never quite made the jump from interesting chemistry experiment to actual heating system in people's homes or businesses. The barriers were always the same: the molecules didn't store enough energy, they degraded too quickly when you tried to reuse them, or they needed toxic solvents that made the whole system impractical.
In early 2026, researchers at UC Santa Barbara and UCLA published a paper that might actually change that. They've engineered a molecule that breaks every record in the MOST space. It stores 1.65 megajoules per kilogram of energy—nearly double what lithium-ion batteries can do. It's a liquid at room temperature, so it doesn't need toxic solvents. It's reusable, at least in the lab. And it was inspired by something unexpected: the molecular damage that UV light causes to DNA.
Let's dig into what makes this breakthrough actually matter, what the physics and chemistry look like under the hood, and whether this is actually the moment MOST finally becomes real.
Why Heat Storage Matters More Than You Think
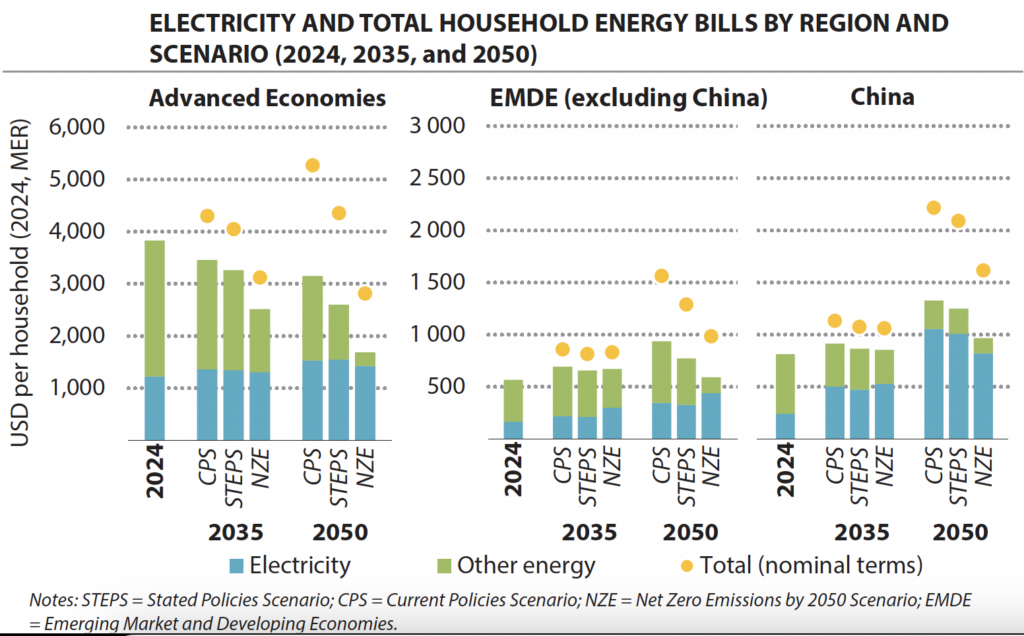
Take a moment to think about your daily energy consumption. The number that usually gets quoted is kilowatt-hours of electricity. But electricity is only part of the story. In most developed countries, residential and commercial buildings use somewhere between 50 and 60 percent of their energy for heating and cooling. In colder climates, heating alone can be 60 to 70 percent of total energy demand. That's not a minor detail you optimize later. That's the core problem.
Industrially, the percentages are even starker. Manufacturing facilities, refineries, chemical plants, paper mills, food processing—they all need consistent, reliable heat. Lots of it. Steel production requires temperatures above 1,600 degrees Celsius. You don't generate that from electricity alone; you generate it by burning something and harvesting the thermal energy. Across the industrial sector, thermal energy accounts for roughly 75 percent of total energy demand.
So when you see statistics about global energy consumption, remember that headline number includes a massive thermal component. And when you see renewable energy growth statistics, those usually track electricity generation. The mismatch matters. You can install solar panels on a roof and generate electricity, but if your building needs heat, you still need something to provide it. And right now, that something is a fossil fuel.
This is why energy companies and climate researchers get excited about thermal storage. A solution that lets you collect solar heat during sunny months and use it during winter? That's not a niche technology. That's a fundamental piece of decarbonizing the economy. It's bigger than electric cars. It's bigger than grid-scale batteries. It's just less visible because it happens inside buildings instead of at charging stations.
There are already some thermal storage solutions in commercial use. Molten salt tanks can store heat for days or weeks. Some facilities use phase-change materials that absorb heat as they melt and release it as they solidify. These work, but they're expensive, require complex engineering, and work best at large scales. For residential buildings or smaller commercial facilities, they're not practical. You need something else. Something that's dense enough to store meaningful energy in a small volume, reusable indefinitely, and compatible with standard heating systems.
That's what molecular thermal storage promises. The energy density of a liquid fuel stored at room temperature, with none of the toxicity or carbon emissions.
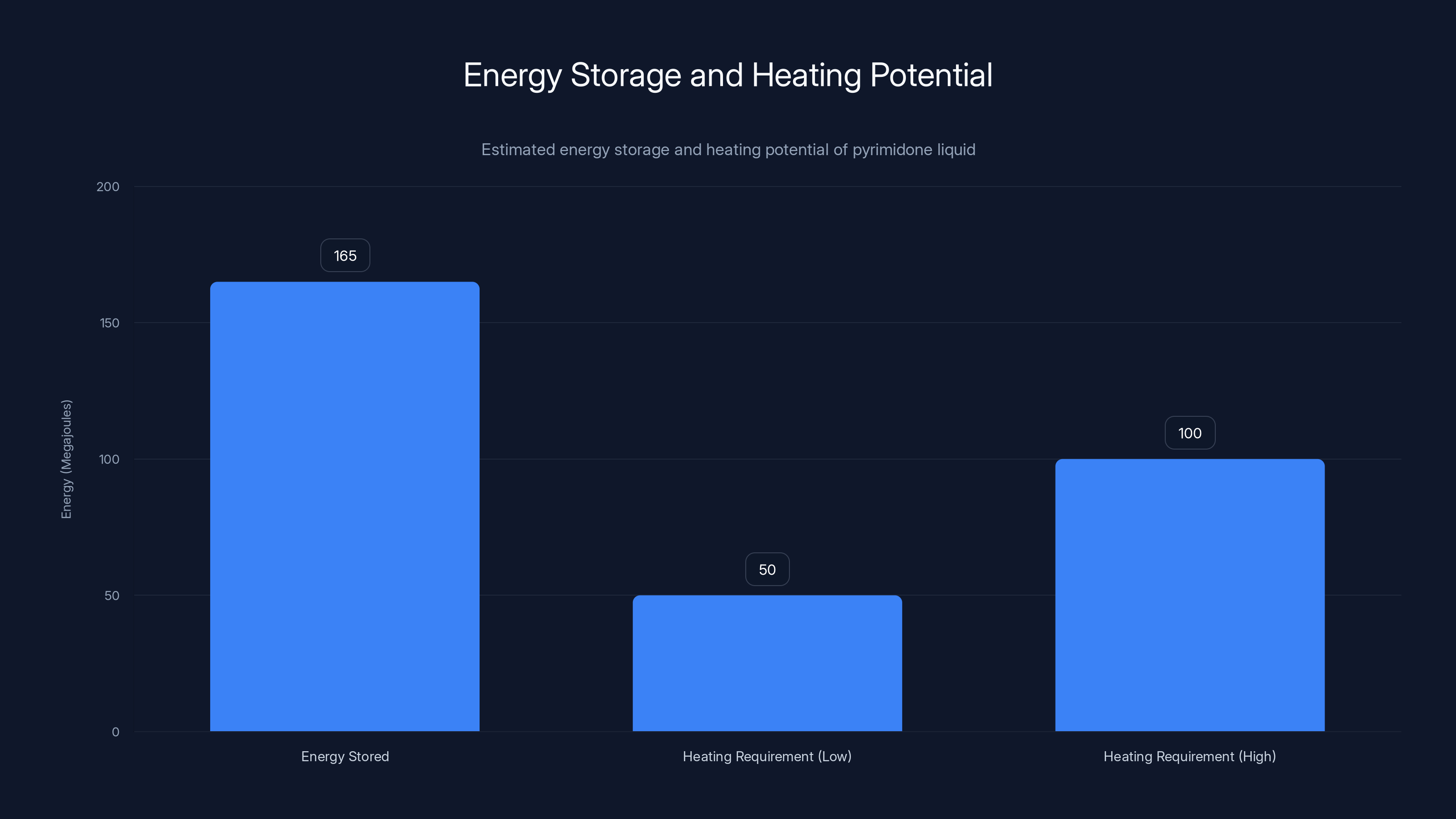
A 100-liter tank of pyrimidone liquid stores 165 megajoules, enough to heat a house for 1-3 days depending on climate and insulation. Estimated data.
The DNA Damage That Inspired a Breakthrough
Sunburn is DNA damage. Specifically, it's UV light hitting the thymine bases in your DNA and causing them to link together. When you spend too long at the beach and get that painful redness, you're experiencing the cellular aftermath of billions of molecules being rearranged at the molecular level.
When two thymine bases sit next to each other and absorb a UV photon, they can form a structure called a (6-4) lesion. Picture it like a covalent bond forming between adjacent letters in a genetic sentence, locking them together. If more UV light hits that lesion, something weird happens. The structure twists into an even stranger configuration called a Dewar isomer. It's a more energetically strained version of the lesion, like taking a piece of the DNA molecule and wringing it out.
In biology, this is bad news. The Dewar isomer creates kinks in the DNA double helix that prevent the molecule from being copied properly. This is how UV light causes mutations and, over time, skin cancer. Your body has evolved specific proteins called photolyases that hunt down these Dewar isomers and snap them back into their original state. It's like a molecular maintenance crew. The bases snap back, the tension releases, and the DNA molecule returns to its relaxed, functional state.
What Han Nguyen's team at UC Santa Barbara realized is that this snap-back mechanism is basically a release valve for stored energy. When a Dewar isomer snaps back to its original form, energy is released. That's heat. And the amount of heat released depends on how much strain was stored in the distorted structure. Dewar isomers are under a lot of strain, so when they snap back, they release a lot of heat.
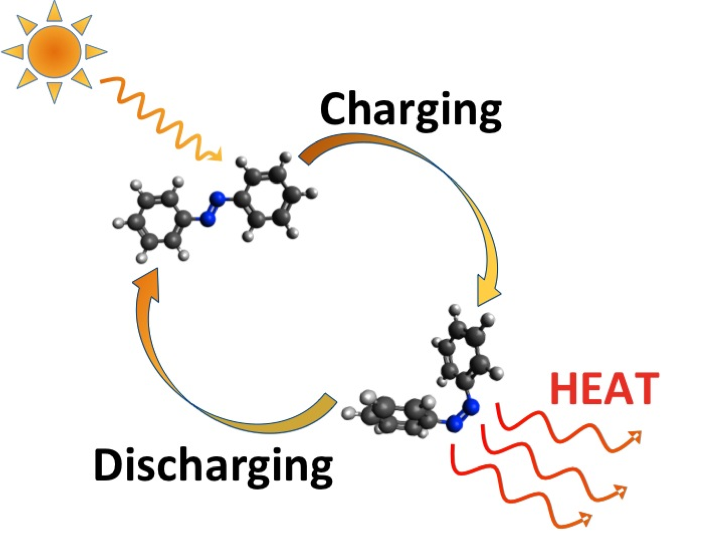
The insight here is subtle but crucial: nature already has a mechanism for storing energy in molecular strain and releasing it on demand. The team decided to engineer a synthetic version—not thymine itself, but a related molecule called pyrimidone—that could do the same thing reliably. Sunlight hits it, it distorts into a highly strained configuration. You want heat, you trigger the release, and it snaps back, dumping all that stored energy.
The elegance of this approach is that you're working with mechanical strain rather than trying to break and remake chemical bonds. You're not burning anything. You're not creating toxic byproducts. You're just taking a molecule from a relaxed state to a strained state and back again, like winding and unwinding a spring. Do it millions of times and the spring should still work, as long as you design it right.
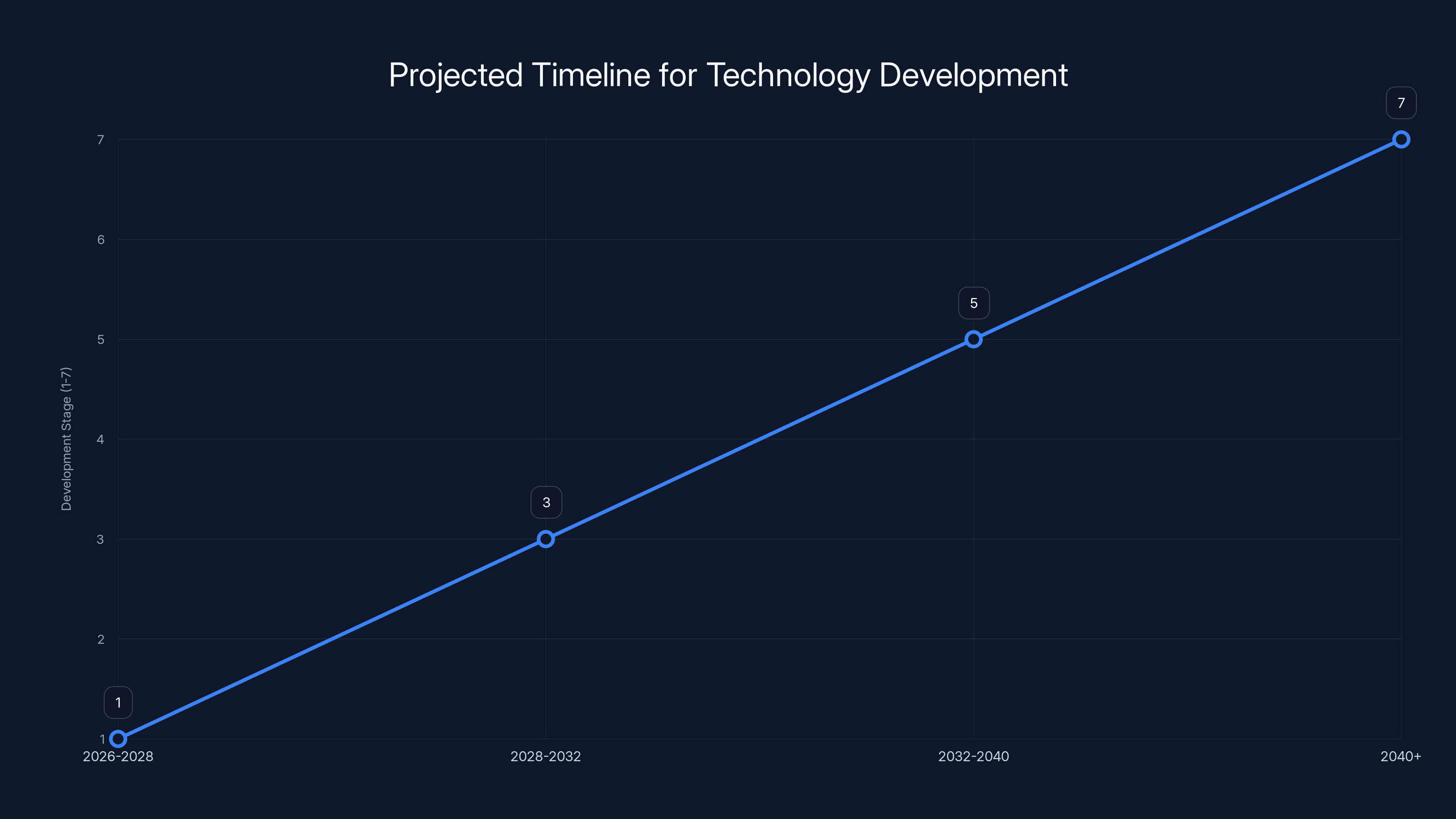
This speculative timeline suggests that technology could move from research to widespread commercial deployment over the next 15+ years. Estimated data.
How the Pyrimidone Molecule Actually Works
Let's get into the actual chemistry, because this is where the breakthrough lives. The team synthesized a molecule based on 2-pyrimidone—one of those chemical cousins of thymine that shows up in all kinds of biological contexts. They engineered it specifically so that when sunlight hits it, the molecule undergoes a predictable transformation. It doesn't just fold slightly. It twists into what's called a fused bicyclic structure containing two different four-membered rings: one called 1,2-dihydroazete and another called diazetidine.
Now, four-membered rings are under enormous structural tension. In chemistry, ring strain is a real thing. A six-membered carbon ring (benzene) is perfectly stable and happy. A four-membered ring is always trying to break apart or rearrange into something more stable. By engineering the pyrimidone so that it forms two of these strained rings fused together, the researchers created a molecule that is energetically desperate to snap back into its original form.
When you shine sunlight on this engineered pyrimidone, the light energy causes an electronic transition that triggers the formation of these strained rings. The molecule absorbs the photon energy and locks it into structural tension. The strained configuration is metastable—it's not falling apart on its own, but it's definitely not happy. It wants to relax. And when you apply the trigger (in this case, an acid catalyst that facilitates the reverse reaction), it snaps back and releases all that stored energy as heat.
The key difference from previous MOST attempts is the amount of strain. Earlier molecules like norbornadiene or azaborinine can store some energy, but the amount is limited by how much strain you can mechanically introduce without making the molecule unstable. The dual four-membered ring design creates compounded strain—it's not just one strained ring, but two of them, amplifying the stored energy. This is why the Nguyen team achieved 1.65 megajoules per kilogram, nearly double previous records.
The numbers matter here. Heating oil, which is ancient solar energy stored in chemical bonds over millions of years, achieves about 40 megajoules per kilogram when you burn it. That's still higher than the new molecule, but remember: you're not burning the pyrimidone. You're using it over and over. A lithium-ion battery stores less than one megajoule per kilogram. The new molecule beats that by a factor of nearly two. For a thermal storage system that doesn't degrade on each cycle, that's unprecedented.
The Liquid State Problem and Why It Matters
Here's where practical engineering enters the picture. Most previous MOST systems rely on solid molecules dissolved in solvents. Toluene. Acetonitrile. Other organic chemicals that can keep the molecule in suspension while allowing photochemical reactions to occur. These solvents are a disaster for energy density. If you dissolve your fuel to 10 percent concentration in solvent, you've just cut your energy density by 90 percent. You're carrying around nine parts water or toxic liquid for every one part actual fuel.
It gets worse. These solvents are not safe to handle. You're not going to pump toluene through pipes in a residential building. If a leak develops, you've got a hazardous spill on your hands. The whole system becomes impractical for real-world deployment. You can build a neat laboratory demonstration, but you can't heat a house with it.
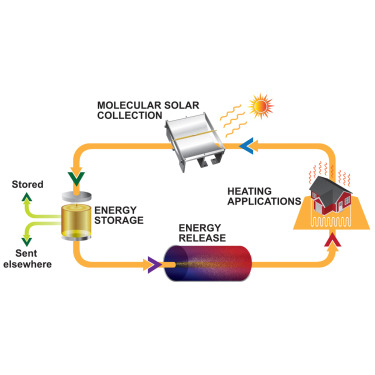
The Nguyen team tackled this by designing their pyrimidone derivative to be a liquid at room temperature. No solvent needed. You just have the fuel itself, flowing as a liquid through solar collectors on the roof, absorbing sunlight, getting charged up, then flowing into a tank in the basement where it waits. Months later, you pump it to a reaction chamber where an acid catalyst triggers the release, and the energy heats a working fluid that goes through a standard heat exchanger and into your building's heating system.
Being a liquid at room temperature means dramatically simpler engineering. You can use standard pumps and piping. You don't need exotic materials or sealed containers. The whole system looks more like a conventional solar heating setup, which means more engineers know how to install and maintain it.
There's another advantage that might seem small but isn't: the molecule is compatible with aqueous environments. It works in water. This might sound trivial, but it means if there's a leak, you're not spraying hazardous chemicals around. The researchers even demonstrated that the molecule can work dissolved in water and that when the energy release reaction occurs, the temperature increase is intense enough to boil the water. For a residential heating system, that's exactly what you want.
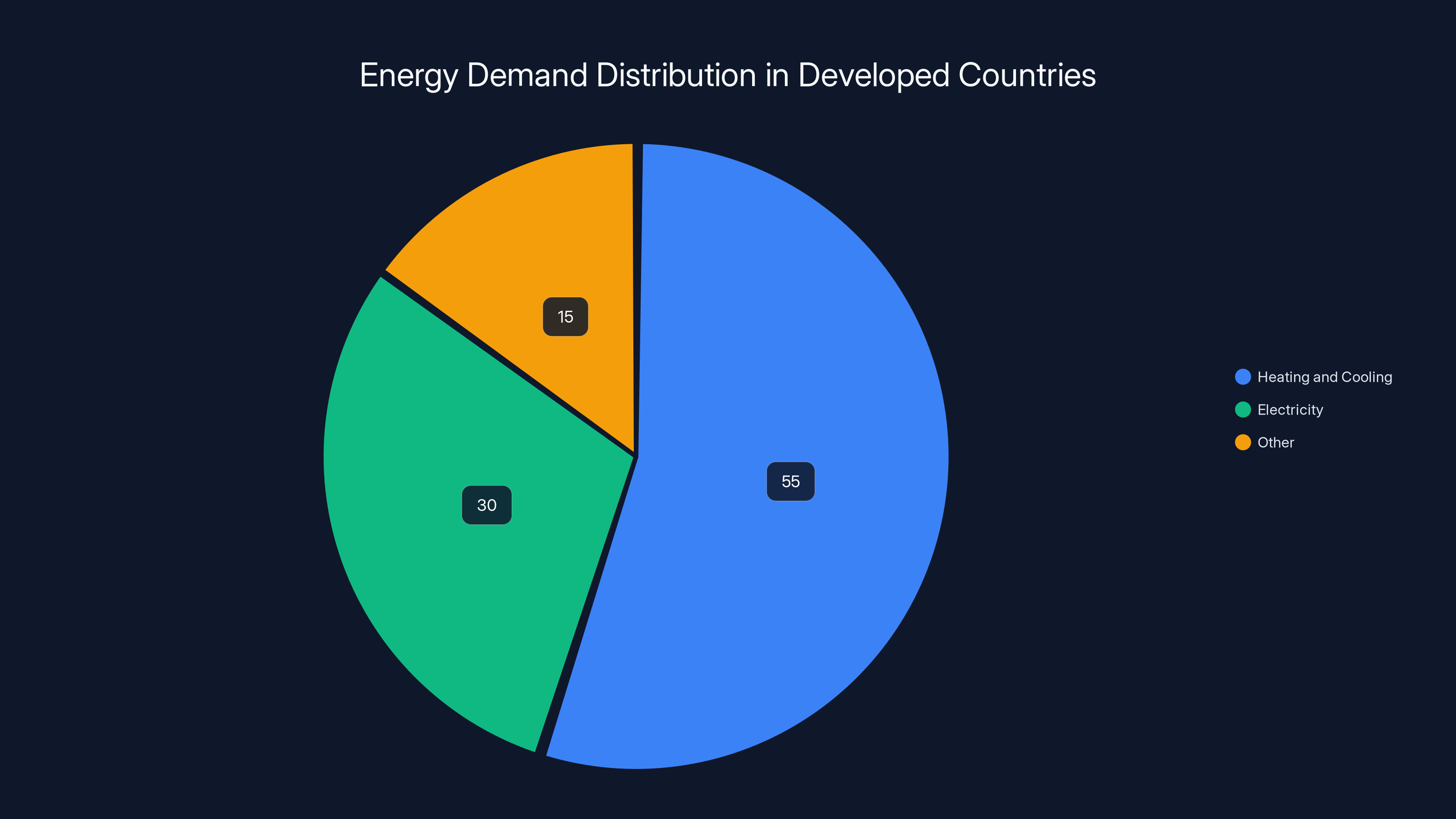
Heating and cooling account for approximately 55% of energy demand in developed countries, highlighting the importance of thermal storage solutions. Estimated data.
Energy Density Compared to Other Storage Technologies
Let's put some numbers on this, because the comparison really highlights what the breakthrough achieved.
Lithium-Ion Batteries: 0.9 to 0.95 megajoules per kilogram. This is what powers most of your electronics and electric vehicles. It's good at storing electricity for hours or days. It's terrible at storing heat for months.
Norbornadiene: 0.97 megajoules per kilogram. This is one of the best-studied MOST candidates from previous decades. Researchers have been optimizing this molecule since the 1990s. It was the reigning champion of the MOST space. It's still worse than lithium-ion.
Azaborinine: 0.65 megajoules per kilogram. Another candidate that attracted serious research funding. It under-performs even norbornadiene.
New Pyrimidone Derivative: 1.65 megajoules per kilogram. More than 70 percent better than the previous MOST champion. 75 percent better than lithium-ion batteries. And remember, this is in the lab. The researchers have not yet optimized the synthesis or the system design. There's room for improvement.
Heating Oil: 40 megajoules per kilogram. Still higher, but you burn it once and it's gone. The pyrimidone can be used thousands of times.
The practical implication: if you have a 100-liter tank of this liquid in your basement, you're storing about 165 megajoules of energy. That's roughly equivalent to 46 kilowatt-hours. For a typical U.S. home, that might cover a day or two of heating in winter, depending on outside temperature and insulation. For a building that needs consistent process heat, you'd scale up the tank size. But the point is that the energy density is finally high enough that the system doesn't require enormous storage volumes.
How a Real-World Heating System Would Work
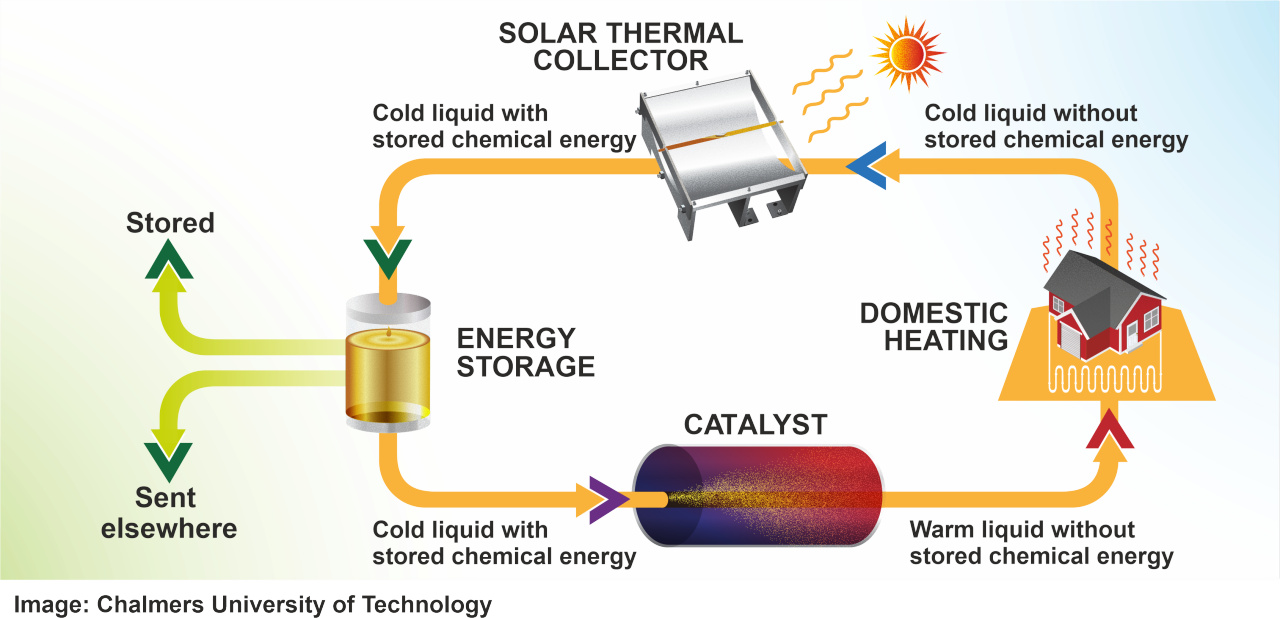
Imagine a residential setup. Solar panels on the roof, but these aren't photovoltaic panels that generate electricity. They're solar collectors, similar to what you'd use for solar hot water, but with a different fluid flowing through them. This fluid is the engineered pyrimidone liquid. During sunny months, this fluid circulates through the roof collectors, absorbs solar energy (converting it to structural strain in the molecules), and then flows down to a tank in the basement or a utility room. The tank is insulated to minimize heat loss, though at room temperature there's minimal radiation anyway.
Months pass. Summer becomes fall. Fall becomes winter. The tank sits there, storing all that bottled solar energy in molecular form. No degradation. No leakage. The molecules don't care if they're stored for a week or four months. They're stable at room temperature.
When you need heat—a cold winter day, or an industrial facility needing process heat—the fluid from the tank gets pumped to a small reaction chamber. This chamber contains an acid catalyst, probably something simple like sulfuric acid or a solid acid resin. The acid triggers the reverse reaction. Those strained bicyclic rings snap back into their relaxed form. The stored energy releases as heat, warming the fluid. This hot fluid then flows through a heat exchanger where it transfers that thermal energy to the working fluid in your building's heating system. The cooled fuel cycles back to the tank, ready to be charged up again next sunny day.
The whole loop is closed. Efficient. No combustion. No emissions. No ash or exhaust. The acid catalyst doesn't get consumed in the reaction—it's regenerative, so you're not adding fresh catalyst every cycle.
For industrial applications, the setup scales similarly but with different numbers. A large facility might have much bigger roof collector areas and much larger storage tanks. The thermal energy might be used directly in a process rather than through a heat exchanger. But the core mechanism is identical.
The elegance is that this system slots into existing infrastructure. Buildings already have heating systems. Industrial facilities already use thermal energy. This technology doesn't require reinventing the wheel. It provides a drop-in replacement for "import heat by burning something" with "import heat from previously captured sunlight."

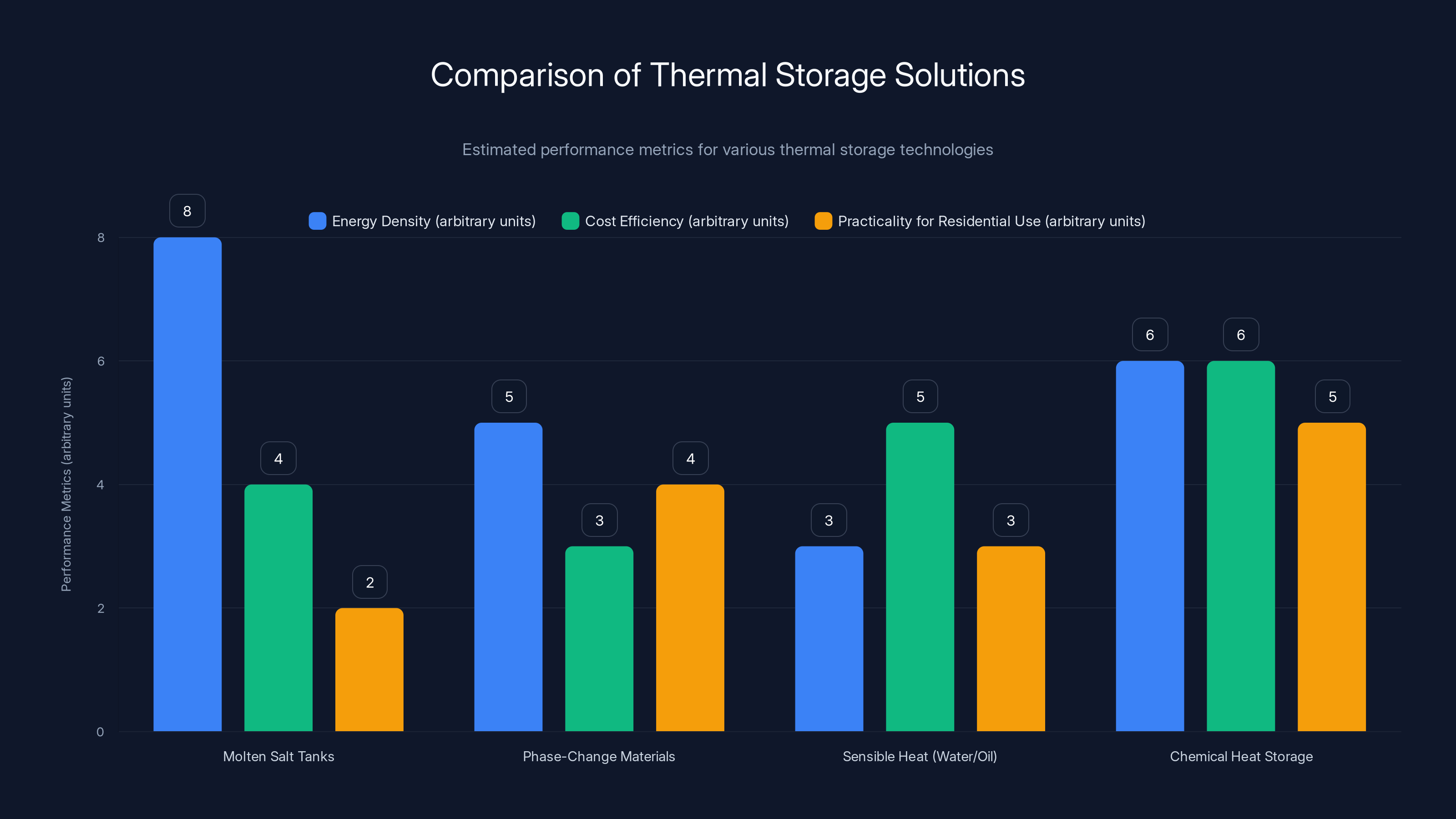
The pyrimidone system offers higher energy density compared to phase-change materials and sensible heat storage, with better practicality for residential use than molten salt tanks. Estimated data.
The Spectral Challenge: What Wavelengths Actually Work
This is where reality gets messier than the laboratory. The sun delivers a broad spectrum of light: infrared, visible, ultraviolet. Different photons have different energies. The engineered pyrimidone absorbs light in a specific range of wavelengths. Not all sunlight is equally useful.
The researchers designed the molecule to absorb primarily in the UV and near-UV range, the same wavelengths that trigger the photolyase reaction in natural DNA. This is where the molecule is most efficient at converting photon energy into structural strain. But the sun's spectrum, especially by the time it reaches Earth's surface, is weighted heavily toward the visible and infrared ranges. UV is actually a small fraction of the total solar energy reaching the ground.
This is a fundamental limitation. You can't make the molecule absorb red light and infrared the same way it absorbs UV without changing its fundamental structure, and changing the structure changes how much energy it can store and how stable it is. So there's a trade-off between spectral efficiency (absorbing as much of the solar spectrum as possible) and energy density (storing as much energy as possible in the absorbed light).
Previous MOST systems faced this same issue. Norbornadiene has similar spectral absorption limitations. It's not a fatal flaw, but it does mean that a real solar collector using this technology wouldn't be 100 percent efficient at converting incident solar energy into stored thermal energy. The theoretical maximum, accounting for the spectral limitations and the Carnot efficiency limits of thermal systems, might be somewhere in the 20 to 30 percent range. That's lower than modern photovoltaic panels (which achieve 15 to 22 percent), but it's comparable to some solar thermal systems.
The researchers note in their paper that there are strategies to address this. You could use dichroic mirrors to preferentially route UV photons to the collector while allowing infrared to pass through for other uses. You could engineer additional chromophores into the system to absorb different wavelengths. You could use the system in climates with high UV intensity. These are all solvable problems, not fundamental blockers.
Stability and Degradation: The Reusability Question
Previous MOST molecules had a serious problem: they didn't last. You'd charge and discharge them a few times, and the molecule would break down or react with something in the solvent or just drift away from the desired reaction pathway. The whole system degraded within weeks. If you can't reuse the fuel indefinitely, the economics don't work. You'd be constantly replacing the expensive engineered molecules.
The Nguyen team tested their pyrimidone derivative for stability. They cycled it through charge-discharge cycles in the lab and monitored what happened. After 100 cycles, there was minimal degradation. The molecules still performed essentially as designed. They didn't degrade into side products or lose their ability to store and release energy.
This is crucial. It's the difference between a laboratory curiosity and an actual usable technology. A home heating system that required replacing its thermal storage fluid every month or two would be economically nonsensical. Replacing it every five or ten years, or even every twenty years, becomes a standard maintenance item, like replacing a water heater. That's acceptable.
But here's the reality check: 100 cycles in a lab over a few weeks is not the same as years of real-world use. Lab conditions are controlled. Temperature is stable. Humidity is managed. The fluid isn't exposed to dust, contaminants, or fluctuating conditions. Real roofing gets hot in summer, cold in winter, bakes under UV radiation. Real pipes corrode or outgas. Real systems have failures you can't predict in advance.
The researchers acknowledge this. They tested the molecule in water and in liquid state and under various pH conditions. They demonstrated it works. But they haven't yet deployed a real system on a real roof for a real winter. The jump from "stable for 100 lab cycles" to "reliable for 20 years in an actual building" is significant. It's not insurmountable—solar hot water systems have made that jump—but it's not automatic either.
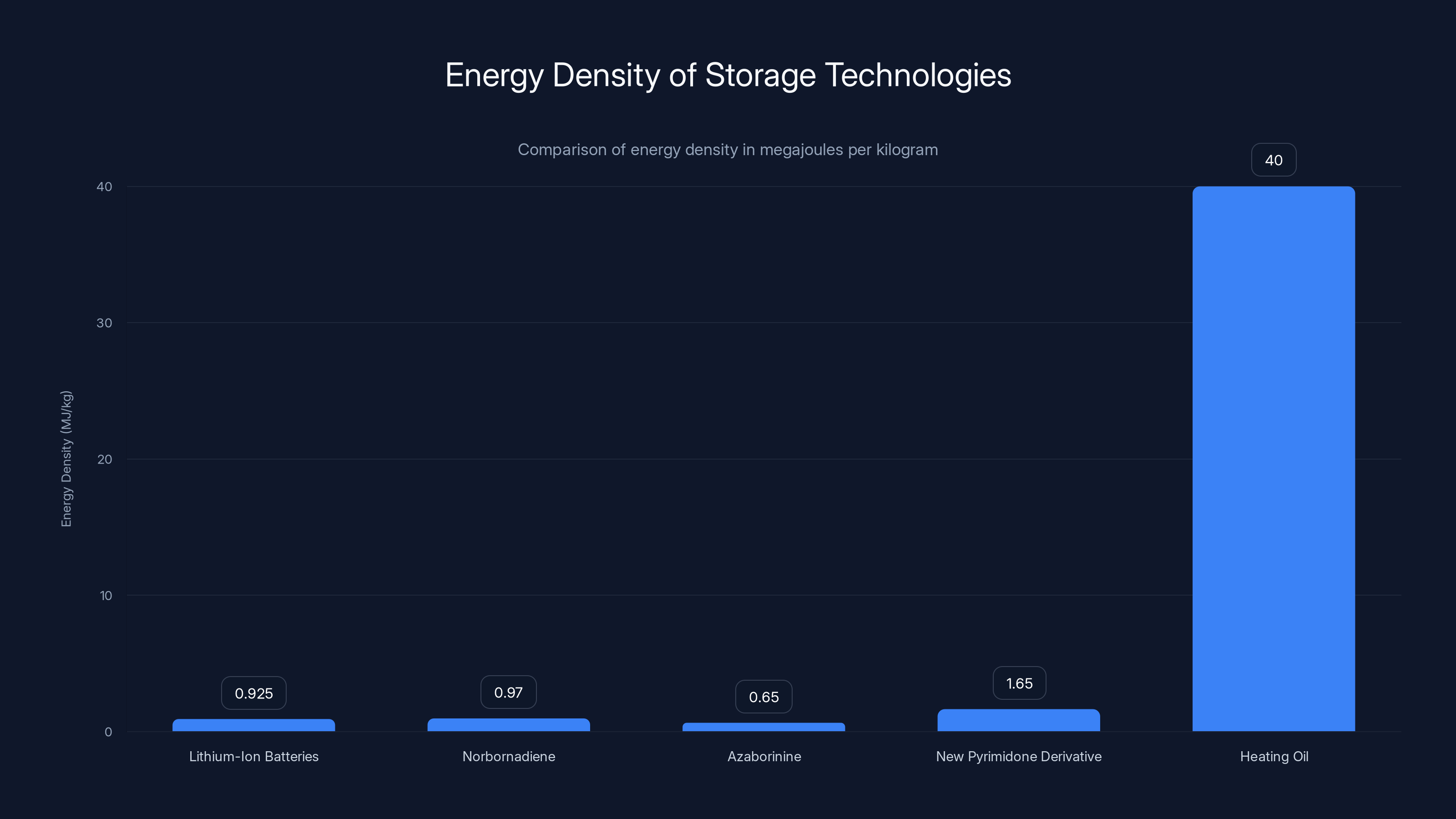
The New Pyrimidone Derivative demonstrates a significant improvement in energy density over previous MOST candidates and lithium-ion batteries, though it still falls short of heating oil. Estimated data.
Trigger Mechanisms and Controlled Release
Storing energy is half the problem. Releasing it on demand is the other half. You don't want the molecule spontaneously flipping back to its relaxed state while sitting in your basement tank. You need a trigger that's reliable, safe, and easy to control.
The team uses an acid catalyst. When the stored liquid is exposed to acid (or to a surface coated with acid), the reverse reaction is triggered. The strained rings snap back, releasing heat. The reaction is energetically favorable once the catalyst is present, which means it goes essentially to completion. You get most of your stored energy out.
Acid catalysis is a well-understood process in chemistry. There are many ways to implement it. You could use a dilute sulfuric acid solution. You could use a solid acid resin that doesn't need to be in the fluid. You could even design the reaction chamber so that the fluid flows over a catalyst-coated surface, triggering the reaction gradually and controlling the heat release rate.
There are advantages and disadvantages to this approach. The advantage is that it's simple, well-understood, and reliable. The disadvantage is that it requires active management. You can't just expose the tank to acid and walk away—you need to control the reaction rate to match your heating demand. This means the system needs some level of automation or at least regular attention.
Compare this to passive thermal storage systems like molten salt, which release heat naturally through conduction without needing a trigger. The pyrimidone system requires more active management. But that's actually fine. Most building heating systems are actively managed anyway. A thermostat controls whether the furnace or boiler is on or off. Adding a control system that regulates when the thermal storage fluid flows to the catalyst chamber is not a huge additional complexity.
The researchers emphasize that the trigger mechanism can be tuned. You could make the reaction faster or slower by adjusting the catalyst concentration or temperature. You could design the reaction chamber for different flow rates. The fundamental point is that the trigger mechanism works reliably, and you have control over when and how much energy is released.
Comparing to Existing Thermal Storage Solutions
There are actually several thermal storage technologies already in use or in serious development. Understanding how the new pyrimidone system compares helps contextualize the breakthrough.
Molten Salt Tanks: These are used at some solar thermal power plants. Salt (usually a mixture of sodium nitrate and potassium nitrate) is heated to over 500 degrees Celsius during the day. At night, the hot salt releases its thermal energy through heat exchangers to generate electricity. The energy density is high, and the storage duration can be days or even weeks. The drawback is that you need extremely high temperatures to make the energy density worthwhile. The salt corrodes pipes. The system requires constant heating to keep the salt liquid, or it solidifies. It's not practical for residential buildings.
Phase-Change Materials: Some compounds melt and solidify at useful temperatures. Paraffin wax, salt hydrates, and other materials can store energy as latent heat of fusion. You heat the material above its melting point during the day; at night, it solidifies and releases that heat. The energy density is decent, but it's still lower than what the pyrimidone achieves. These materials are being explored for building walls and floor systems. They work, but they're expensive and the performance gains are modest.
Sensible Heat in Water or Oil: Heating water or oil and storing it in insulated tanks is the oldest trick in the book. Heat a big tank of water during the day, use it at night. It works but requires enormous storage volumes for anything beyond a few hours of storage. To store enough heat for months, you'd need tanks so large they're economically impractical for residential use. Industrial facilities sometimes use this approach because they have space and capital.
Chemical Heat Storage (Sorption Materials): Some materials, like zeolites or silica gels, can absorb water vapor and release heat when that water is driven off. The reaction is reversible—you can add water back and absorb heat. These systems work at moderate temperatures and have decent energy densities. Research is ongoing. Several companies are commercializing these systems for building heating. They work. They're not yet mainstream because of cost and complexity, but they're closer to market than MOST historically has been.
Molecular Solar Thermal (MOST) with Previous Candidates: Norbornadiene, azaborinine, and other molecules achieve lower energy densities than the new pyrimidone but higher than sensible heat storage. These have been researched for decades. They work in lab. They haven't been deployed at scale because of solvent compatibility issues, stability concerns, and the need for more engineering development.
The new pyrimidone system potentially combines the best traits: high energy density (better than most alternatives), liquid state (simpler engineering than solids), reusability (better than one-shot combustion), and room temperature stability (simpler than molten salt requiring heating). It's not a clear knockout across every dimension, but it's competitive or superior on most metrics.
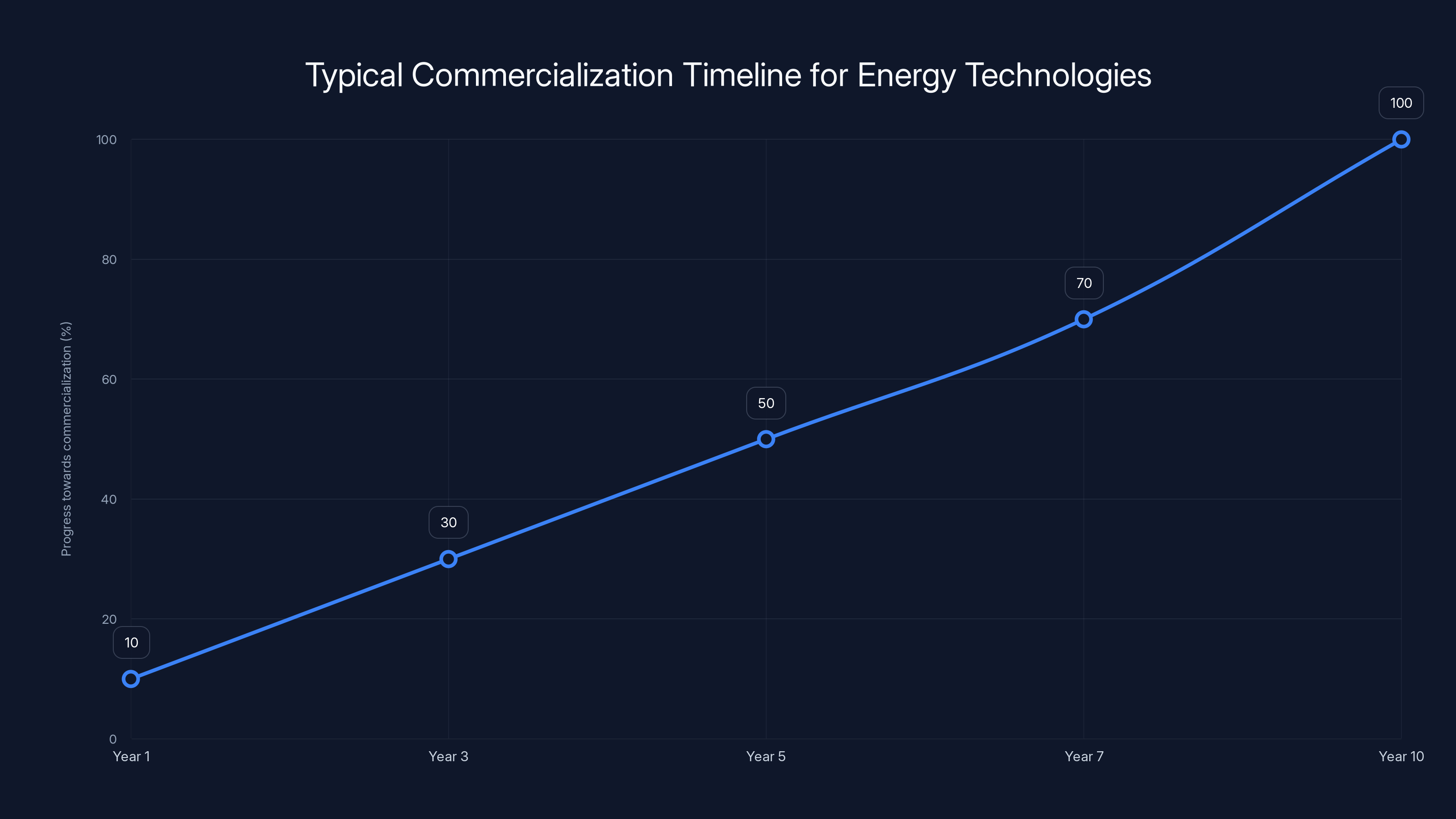
The transition from lab to commercial deployment for energy technologies typically spans 5-10 years, with incremental progress in development, testing, and deployment. Estimated data.
The Path to Commercialization: From Lab to Roof
Here's the honest assessment: a breakthrough paper in Science is not the same as a product you can buy. The transition from "we demonstrated this in a well-funded laboratory at a top university" to "this is reliably heating buildings across the country" involves years of additional work.
The researchers have demonstrated the concept and achieved the performance metrics in a carefully controlled lab environment. They've measured energy storage and release. They've cycled the molecule repeatedly. They've tested it in water and in liquid form. They've shown that the reaction is triggered reliably by acid catalysis. All of this is real and reproducible.
But there are still questions. How does the molecule perform over years or decades of exposure to real conditions? How much does it cost to synthesize at scale? What's the manufacturing process? How do you handle the spent fluid—is it recycled, treated, disposed? What safety approvals are needed for a system that uses acid as a trigger? How do you design the collector panels? What's the whole-system efficiency when you account for pump energy, heat exchanger losses, and storage tank heat loss? How long until the first real building uses this for actual heating?
These are not trivial questions. They're standard engineering questions, but they require time, funding, and iterative development. Some companies are probably already interested. Universities might spin out a startup. A larger company in the thermal energy space might license the technology. But the real proof will come when someone actually builds a system and deploys it and runs it through a winter and reports back on how well it works.
That transition typically takes five to ten years, sometimes longer. Molten salt systems were researched in the 1970s and didn't reach commercial deployment until the 2000s. Phase-change materials have been in research for thirty years and still haven't achieved widespread deployment. Sorption-based systems are just reaching the prototype stage now.
The point isn't to be pessimistic. The point is to be realistic. The science is genuinely exciting. The performance metrics are genuinely impressive. But between impressive lab results and "your home was heated by solar energy captured six months ago" is a gap that takes years and millions of dollars to cross.
Cost Economics and Market Potential
Let's think about the economics. Suppose you have a 100-liter tank of this pyrimidone liquid. That 100 liters stores about 165 megajoules of energy. To heat a typical house from 60 degrees Fahrenheit to 70 degrees for a full day in winter (accounting for thermal losses), you might need 50 to 100 megajoules. So your 100-liter tank could heat a house for one to three days, depending on climate and insulation.
To actually heat a house all winter (say, six months in a cold climate), you'd need either very large roof collector panels to charge the tank multiple times over the winter, or you'd need multiple storage tanks, or you'd need a hybrid system where MOST handles some of the heating and a backup system handles the rest. This gets complicated quickly and depends heavily on local climate and building specifics.
The economics depend on several things: how much the pyrimidone costs to manufacture, how efficient the collector panels are, how much the heat exchanger and pump systems cost, how long the whole system lasts before needing replacement, and what you're replacing. If you're replacing a natural gas furnace in a place where natural gas is cheap, MOST has a harder time competing. If you're in a place where heating oil is expensive or where electricity rates are high, MOST looks better.
Right now, the cost structure is probably not favorable for residential deployment at scale. The molecules are lab-synthesized. Manufacturing costs haven't been optimized. The system hasn't been engineered for mass production. First-generation systems will be expensive. If the technology matures, costs will come down. Solar photovoltaic costs dropped by 90 percent over two decades. Lithium-ion battery costs dropped by 95 percent over a decade. Similar cost curves are possible for MOST if the technology reaches scale.
For industrial applications, the economics might be better sooner. Large facilities with high thermal energy demands in sunny climates could potentially benefit from a system that stores seasonal heat, even if the system is expensive. A chemical plant or brewery or food processor using MOST for process heat could eliminate natural gas and improve their carbon footprint. The payback period might justify the initial capital investment.
There's also a policy dimension. If carbon pricing becomes more aggressive, or if natural gas becomes more expensive or constrained, the economics shift in favor of MOST. If governments subsidize solar thermal systems the way they've subsidized solar PV and heat pumps, adoption would accelerate.

Broader Implications for the Energy Transition
Thermal storage is a missing piece in the clean energy puzzle. We've made real progress on electricity generation (renewables are now cheaper than fossil fuels in most places) and on electricity storage (battery costs keep falling). But the thermal piece has been neglected. Heating and cooling are still deeply dependent on fossil fuels and will remain so until we have better thermal storage solutions.
A working MOST system changes the calculus. Suddenly, seasonal heat storage becomes feasible. A building in a Mediterranean climate could store summer heat in a basement tank and use it for winter heating. A building in a continental climate with extreme winters and sunny summers could do the same. Industrial facilities could smooth out demand and use renewable energy more efficiently.
This is particularly important in northern latitudes where heating demand is highest and sunshine is most seasonal. Much of Europe, Canada, northern United States, and Scandinavia struggle with the mismatch between peak heating demand (winter) and peak solar availability (summer). A technology that bridges that gap doesn't just provide an alternative to natural gas. It fundamentally changes the architecture of the energy system.
Beyond heating, there are other applications. Concentrated solar thermal power plants (CSP, or solar thermal electricity) could use MOST for thermal storage instead of molten salt, potentially achieving higher energy densities and lower costs. Thermal storage could integrate better with high-temperature industrial processes. Even data centers, which generate massive waste heat, could potentially use MOST systems to store and recover that heat for other uses.
The broader point: if MOST becomes practical and cost-competitive, it opens up entire categories of applications and system designs that aren't feasible with current technology. It's not just an incremental improvement on existing thermal storage. It's a different approach that could enable genuinely new ways of managing energy across the economy.
Remaining Technical Challenges and Research Directions
The Nguyen team has demonstrated proof of concept. But there are still open questions that future research will need to address. Some are materials science questions. Some are engineering questions. Some are economic or policy questions.
Optimization of the Molecule: The current pyrimidone derivative is good, but is it optimal? Could you engineer a variant that absorbs a wider spectrum of light? Could you improve the charge-discharge cycling further? Could you make the trigger mechanism even more efficient? Molecular design is an iterative process. The first working version is rarely the best version.
System Integration: Real buildings are complicated. You'd need to design collector panels that work on roofs, tankage that fits in basements or utility rooms, heat exchangers that integrate with existing heating systems, control systems that manage the charging and discharging. This sounds straightforward but involves hundreds of engineering decisions and trade-offs.
Durability and Field Testing: The lab has cycled the molecule 100 times. Real systems need to cycle thousands of times over decades while exposed to dust, UV radiation, temperature fluctuations, and inevitable contamination. Building a field test system and running it through multiple winters would generate invaluable data. Several research groups are probably planning this already.
Scalable Synthesis: Lab synthesis of fine chemicals is expensive and complex. Industrial synthesis requires different approaches, different solvents, different purification methods. Getting the cost per kilogram down from hundreds of dollars to tens of dollars (or ideally single digits) is essential for commercial viability. This is often the hardest part of taking a lab chemical to a commercial product.
Material Compatibility: The fluid needs to work in pipes, pumps, heat exchangers, and storage tanks. Some materials will corrode or degrade in contact with this fluid. Others will remain stable for decades. Identifying and qualifying materials is a standard but essential part of engineering a real system.
Safety and Regulatory: An acid catalyst in a home heating system raises questions. What's the risk of leaks or spills? What happens if someone comes into contact with the fluid? What certifications and approvals are needed? These are solvable but require attention.
Timeline and Future Outlook
If I had to guess—and this is educated speculation, not insider knowledge—here's a plausible timeline.
Next 2-3 years (2026-2028): Follow-up research papers optimizing the molecule, testing longer cycling, demonstrating the system at larger scales. Probably one or two companies or research groups build a real field test system and report results. The technology begins getting attention from energy companies and venture investors.
3-7 years (2028-2032): Prototype systems deployed in real buildings or industrial facilities, probably in Europe or other regions with strong climate policies and higher fossil fuel costs. Initial cost data becomes available. Manufacturing processes improve, costs start coming down. Patent landscape becomes clearer as companies and universities file for protection.
7-15 years (2032-2040): If the technology proves durable and cost-effective, early commercial deployment in favorable markets. Probably starts with large commercial or industrial customers before scaling to residential. Costs continue falling as manufacturing matures. Integration with other renewable technologies (heat pumps, solar PV) becomes common.
15+ years: Mature technology with multiple manufacturers, widespread deployment in suitable climates, cost-competitive with fossil fuel heating in most developed regions. Becomes a standard component in new building designs.
This timeline assumes everything works out. Technology development doesn't always cooperate. Materials that work in the lab sometimes fail in the field. Economics that look promising sometimes don't materialize. But the trajectory is clear: if the core science holds up (and the evidence suggests it will), commercialization is plausible within a decade or two.
Compare this to where we were with lithium-ion batteries in 2005. The technology existed but was expensive and not widely used. Fifteen years later, lithium-ion was in phones, electric vehicles, and grid-scale storage systems. The path from lab to ubiquity usually takes something like that timeline. MOST is in the 2005-2010 stage now. The breakthrough is real, but the journey to commercialization is just beginning.
The Bigger Picture: Beyond MOST
Thermal storage is important, but it's one piece of a larger puzzle. The clean energy transition requires not just renewable electricity, but also renewable thermal energy, sustainable transportation, sustainable materials, and sustainable agriculture. Each of these has its own technical challenges and its own breakthroughs needed.
For thermal energy specifically, MOST is one approach among several. Sorption systems are another. Heat pumps are another (though they're more about moving heat than storing it). Different solutions will work best in different contexts. A building in a sunny desert climate might use MOST. A building in a cloudy, temperate climate might use a heat pump backed up by renewable electricity. A factory might use industrial waste heat recovery. The ultimate energy system will probably use all of these tools in combination.
But MOST is significant because it directly tackles a fundamental problem: seasonal mismatch between energy supply and demand in a renewable energy system. It's not a minor optimization. It's a foundational technology for making deep decarbonization feasible. If it works and becomes cost-competitive, it unlocks possibilities that aren't feasible today.
The other thing worth noting: this breakthrough came from a team at UC Santa Barbara, a publicly funded university, publishing their results in a top-tier journal. The research was probably funded by the Department of Energy or the National Science Foundation or both. This is basic research that adds to human knowledge and capability. Even if this particular version of MOST never makes it to commercial deployment, the insights will contribute to other work in thermal storage, molecular engineering, and energy science.
Science is often described as a straight path from discovery to application. In reality, it's messy. Hundreds of papers are published. Most lead nowhere. A few lead somewhere. The ones that lead somewhere usually succeeded because researchers built on the work of others, stood on the shoulders of giants, adapted old ideas to new contexts. The DNA inspiration for this breakthrough is a perfect example. Natural mechanisms inspire engineered solutions. Biology suggests chemistry and materials science can do things we hadn't thought to try.
Conclusion: When Laboratory Breakthroughs Become Real-World Solutions
Let's zoom out and think about what this research actually means.
For the first time, a team has engineered a molecule that stores solar energy at unprecedented density, in a safe liquid form, without requiring toxic solvents, with demonstrated reusability, and with clear pathways for integration into existing heating infrastructure. That's a genuine breakthrough. It solves multiple problems that previous MOST systems failed to address.
But breakthrough papers and deployable systems are not the same thing. The research is real. The results are impressive. The potential is enormous. But between here and there is years of additional work: optimization, system integration, field testing, cost reduction, regulatory approval, and market development. The researchers haven't solved every problem. They've solved the core technical problem that was preventing MOST from working. The remaining problems are real but are the kind of problems that engineering teams and companies and manufacturing plants know how to solve.
So where does this leave us? If I'm being honest: cautiously optimistic. The science is solid. The approach is elegant. The energy density is genuinely high. The safety and practicality are far better than previous attempts. These are all prerequisites for a technology to eventually reach real-world deployment. But prerequisites are not the same as guarantees. The next five years of follow-up research, field testing, and prototype development will determine whether this is genuinely the breakthrough it appears to be, or whether it's one of dozens of laboratory-stage innovations that never quite make it to market.
What I'm confident about: heating and thermal storage matter far more than they usually get credit for. If you're tracking the clean energy transition, you should be watching thermal storage technology as closely as you watch battery technology. They're equally important. Second, DNA-inspired molecular engineering is a powerful approach that will probably yield more breakthroughs in the coming years. Using biology as a source of inspiration for engineering—that's not new, but it's being applied in more sophisticated ways. Third, the researchers at UC Santa Barbara and UCLA have done something real and valuable. Whether it becomes a world-changing technology or a stepping stone toward other technologies, the contribution is legitimate.
The heat problem is fundamental. The solution might not be this specific molecule. But we need some solution to make deep decarbonization feasible. A decade ago, if you'd asked engineers and energy researchers what the biggest unsolved problems were, thermal storage would have been near the top. We had electricity pretty well figured out. We were working on transportation. But heat—months-long seasonal heat storage—was intractable. Now we have a molecule that might make it tractable. That's worth paying attention to.
FAQ
What is molecular solar thermal energy storage?
Molecular solar thermal (MOST) energy storage is a technology that uses specially engineered molecules to capture and store solar energy in chemical bonds, then releases that energy as heat on demand. When sunlight hits these molecules, they undergo a structural transformation that stores the energy in mechanical strain. Later, when heat is needed, a trigger (like an acid catalyst) causes the molecules to snap back to their original form, releasing the stored energy as heat. Unlike batteries that store electricity, MOST stores thermal energy directly, which is useful for heating buildings and powering industrial processes.
How does the new pyrimidone molecule differ from previous MOST candidates?
The new pyrimidone-based molecule achieved several breakthrough improvements over previous MOST systems. First, it stores 1.65 megajoules per kilogram of energy—nearly double the capacity of previous molecules like norbornadiene (0.97 MJ/kg) and far superior to azaborinine (0.65 MJ/kg). Second, it's a liquid at room temperature, eliminating the need for toxic solvents that previous systems required. Third, it's compatible with aqueous environments, meaning leaks aren't hazardous. Finally, the engineering team demonstrated that the molecule can be cycled repeatedly through charge-discharge cycles without significant degradation, suggesting it could be reused indefinitely rather than being burned like fossil fuels.
Why does thermal energy storage matter more than people realize?
Heating accounts for nearly half of global energy demand worldwide, and roughly two-thirds of that is currently provided by burning fossil fuels. Yet when discussing the clean energy transition, most attention focuses on electricity generation and storage. This creates a fundamental imbalance: we've solved renewable electricity reasonably well, but we haven't solved renewable heating. A technology that lets you capture solar heat in summer and use it in winter addresses one of the biggest unsolved problems in decarbonization. For northern climates with extreme seasonal variation, thermal storage is essential to achieving net-zero emissions without relying on fossil fuels or electricity heating.
What is the energy density compared to other technologies?
The new pyrimidone molecule achieves 1.65 megajoules per kilogram, which is substantially higher than previous MOST materials and comparable to lithium-ion batteries (which store less than 1 MJ/kg). While heating oil still stores more energy per unit mass (about 40 MJ/kg), heating oil can only be used once, then it's gone. The pyrimidone can be cycled thousands of times, making its long-term effective energy value much higher. The high energy density means practical storage systems don't require enormous tank sizes, making residential and commercial deployment feasible.
How would a real heating system using this technology work?
A practical system would have solar collector panels on the roof (similar to existing solar thermal systems) through which the liquid pyrimidone flows. As sunlight hits the fluid, it gets charged—the molecules absorb the energy and transition to a strained, higher-energy state. The charged fluid then flows to an insulated storage tank in the basement. Months later, when heat is needed, the fluid is pumped to a small reaction chamber where an acid catalyst triggers the molecules to snap back to their original form, releasing the stored energy as heat. This heat is then transferred through a heat exchanger to your building's standard heating system, the same way current solar hot water systems work.
What challenges remain before this technology can be deployed commercially?
While the breakthrough is genuine, several challenges remain. The molecule needs to be synthesized at industrial scale at reasonable cost, which requires different manufacturing techniques than laboratory synthesis. The system needs real-world field testing through multiple winters to ensure long-term durability under actual conditions. The spectral absorption range is limited to UV and near-UV wavelengths, so not all of the sun's energy can be captured—this is a fundamental property of the molecule design. Regulatory approvals and safety certifications will be needed for residential deployment. And economic modeling needs to show the system can compete with natural gas heating, heat pumps, or other alternatives in various markets. These are solvable problems, but they require years of additional research and development.
When might this technology be available for home heating?
Based on historical timelines for energy technology development, realistic commercialization would likely take 10-20 years. The next 2-3 years will probably focus on follow-up research and prototype field testing. Industrial and large commercial applications might reach early deployment in 5-7 years if funding and development accelerate. Widespread residential adoption would likely follow another 5-10 years after that. For context, molten salt thermal storage was researched starting in the 1970s but didn't reach commercial deployment at scale until the 2000s. The timeline depends heavily on funding, policy support, manufacturing investment, and whether field testing confirms the laboratory results.
Could this approach be used for industrial heating processes?
Yes, industrial applications might actually be more promising near-term. Large facilities with high thermal energy demands—chemical plants, breweries, food processors, paper mills—could use MOST systems to provide process heat without burning fossil fuels. Industrial settings often have space for larger storage tanks and can justify higher capital costs if the payback period makes sense. These applications might drive down manufacturing costs and operational experience faster than residential systems, eventually making the technology viable for homes as well.
How does this discovery fit into the broader clean energy transition?
Thermal storage is a foundational technology for deep decarbonization, yet it's received far less attention and investment than electricity storage or renewable electricity generation. By enabling seasonal heat storage—capturing solar energy in summer and using it in winter—MOST technology addresses a fundamental mismatch between energy supply and demand in renewable systems. This is particularly critical in northern climates with extreme seasonal variation. If this technology matures and becomes cost-competitive, it could enable nearly complete decarbonization of heating, which is essential for reaching net-zero emissions without relying on electricity heating or biomass.
What inspired the DNA-based approach to this molecule design?
The researchers drew inspiration from the molecular damage caused by UV light in sunburn. Specifically, when UV light hits adjacent thymine bases in DNA, they can link together and form structures like Dewar isomers, which are highly strained and unstable. Evolution created enzymes called photolyases that snap these strained structures back to their original form, releasing energy as heat in the process. The team realized this mechanism—storing energy in molecular strain and releasing it on demand through a snap-back reaction—could be engineered into synthetic molecules for thermal storage. Rather than trying to store energy in the breaking and making of chemical bonds (like combustion), they engineered the pyrimidone to store energy in mechanical strain, then designed it to snap back on command using an acid trigger.
Key Takeaways
- It's a liquid at room temperature, so it doesn't need toxic solvents
- These work, but they're expensive, require complex engineering, and work best at large scales
- It's like a molecular maintenance crew
- Do it millions of times and the spring should still work, as long as you design it right
- The key difference from previous MOST attempts is the amount of strain
Related Articles
- NYT Strands Answers & Hints Game #716 Feb 17 [2025]
- PS5 Presidents' Day Deals 2025: $100 Off + Fortnite Bundle [2025]
- 2026 TechRadar Australian PC Awards Finalists: Complete Guide [2025]
- Best Budget Camera Lenses for Sony, Nikon & Fujifilm [2025]
- Hands-on: Acer Veriton GN100 AI mini PC workstation | TechRadar
- Best 4K Blu-ray Starter Packs for Ultimate Movie Quality [2025]
![Solar Thermal Energy Storage: How Molecules Store Heat for Months [2025]](https://tryrunable.com/blog/solar-thermal-energy-storage-how-molecules-store-heat-for-mo/image-1-1771265418450.jpg)