Electrides: The Mysterious Materials Hiding Earth's Missing Elements
For nearly a century, Earth scientists have been chasing a ghost. When they look at the chemical composition of our planet compared to the Sun and pristine meteorites, something doesn't add up. We're missing a lot of stuff. I'm talking about hydrogen, carbon, nitrogen, sulfur, and noble gases like helium. In some cases, we're short by more than 99 percent. The question that keeps geoscientists up at night is simple but profound: where did all these elements go?
You might think the answer is obvious. Maybe they floated away into space during Earth's formation. That explanation handles some of the loss, sure. But researchers have suspected for decades that there's another mechanism at work, something deeper and stranger. A team of scientists recently proposed a genuinely wild answer: these elements are being absorbed and trapped deep inside Earth's solid inner core, locked away in a form of iron that behaves unlike anything you'd expect from a normal metal.
That exotic form is called an electride, and it's one of the most fascinating chemical oddities scientists have discovered in recent years. But here's the thing that makes this story compelling. Electrides aren't just theoretical curiosities hiding in the planet's center. They're real materials that scientists can now create in laboratories at room temperature. And they're already changing how we manufacture some of the most important chemicals on Earth.
The convergence of planetary mystery and practical chemistry is what makes electrides so captivating right now. You've got researchers using quantum mechanics to understand why Earth's missing elements might be hiding in the core, while simultaneously, chemists are designing new electrides that could make producing fertilizer and pharmaceuticals cleaner and cheaper. This is a story about how understanding the extreme conditions at the center of our planet might help us build a more sustainable future.
TL; DR
- Electrides are exotic materials where electrons occupy spaces between atoms rather than orbiting atomic nuclei, giving them unique electrical and chemical properties.
- Earth's missing elements (hydrogen, carbon, nitrogen, sulfur) may be trapped in electride iron at the core under 360 gigapascal pressure, explaining a mysterious 5-8% density deficit.
- Room-temperature electrides can now be synthesized from multiple elements and are being used to catalyze ammonia production with 20% less energy than traditional methods.
- Catalytic applications are emerging in pharmaceutical manufacturing and green chemistry, with potential to revolutionize how we produce fertilizers and drugs.
- Key challenge ahead is discovering new electride materials and determining the chemical rules governing their formation.

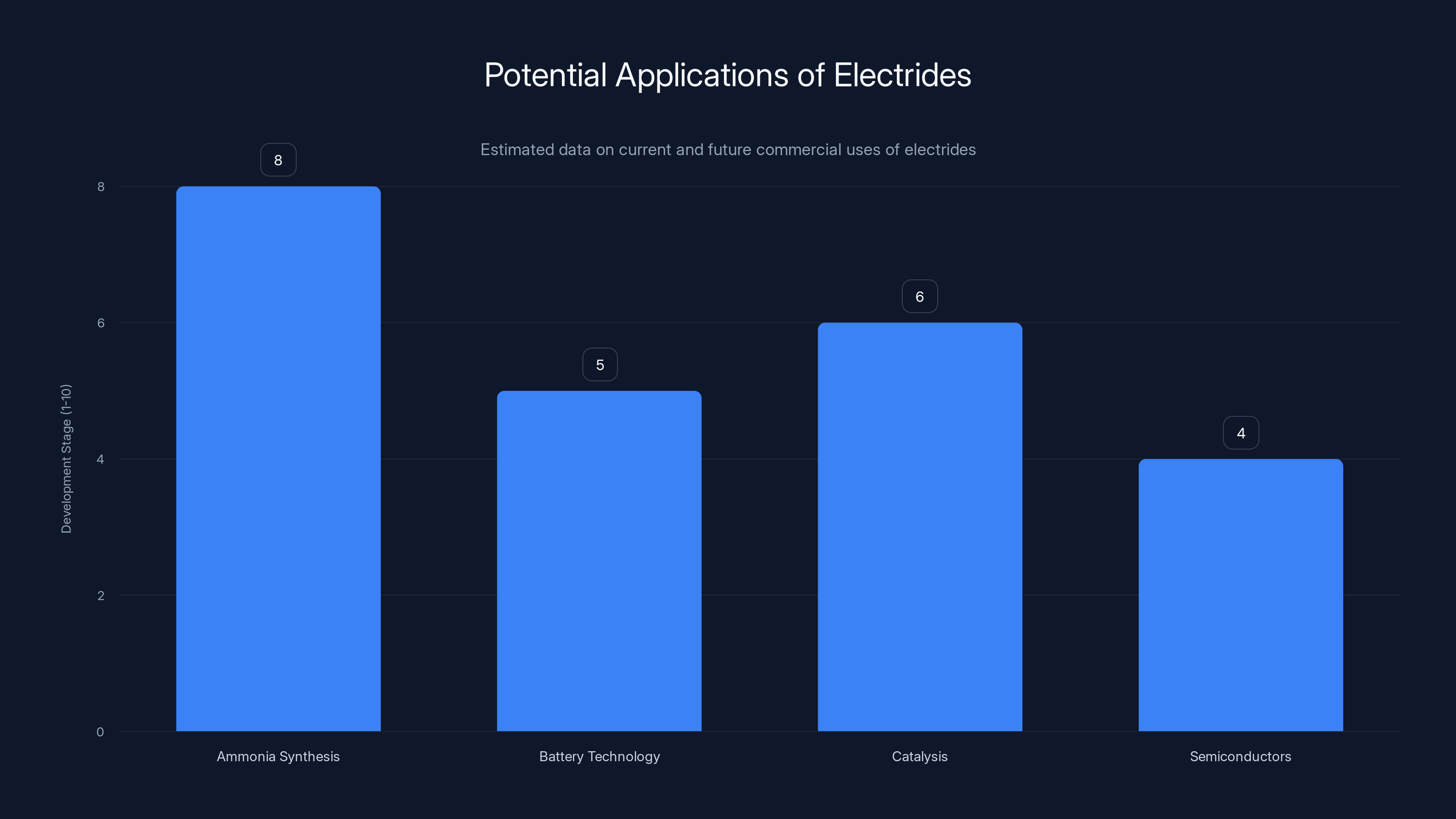
Electrides are most advanced in ammonia synthesis, with significant potential in battery technology and catalysis. (Estimated data)
What Are Electrides, Exactly?
When you think about metals, your mental picture probably involves something pretty straightforward. Atoms packed together in a regular lattice, with electrons flowing freely between them like a liquid. That electron mobility is what makes metals conduct electricity and heat so well. It's why copper wires carry electricity through your house and why aluminum conducts heat in your car's radiator.
Electrides throw this familiar picture out the window. They still have atoms arranged in an ordered lattice, but something strange happens to the electrons. In a normal metal, those outer electrons (called valence electrons) aren't tied to any particular atom. They exist in what chemists call a delocalized sea, free to move around and carry charge. But in an electride, the situation is fundamentally different.
Instead of freely roaming around, the electrons in an electride get trapped at specific locations between atoms. These locations are called non-nuclear attractors, which is a fancy way of saying they're spots in the crystal structure where electrons feel comfortable hanging out, even though there's no atomic nucleus there to anchor them directly. The electrons are held in place by the electrostatic attraction of nearby atomic nuclei, but they can't move freely the way they do in normal metals.
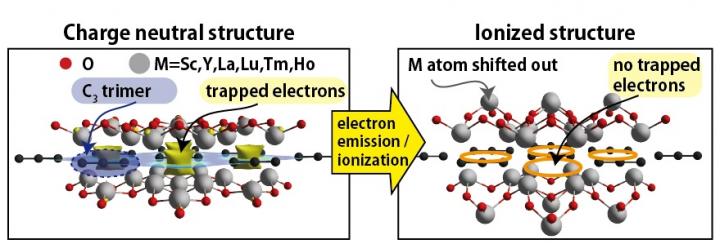
This difference has huge consequences. An electride doesn't behave like a normal conductor. In fact, when sodium transforms into an electride under extreme pressure, it stops being a shiny, reflective metal and becomes something that looks like transparent glass. It loses its metallic luster and becomes an insulator instead of a conductor. The first scientist to observe this transformation in 2009 was absolutely shocked. Sodium going transparent? Under what physical law does that happen?
The discovery of sodium electrides came from computational predictions made in the late 1990s. Theorists were using quantum mechanics to model what happens to solids under extreme pressure. They discovered that as you squeeze atoms closer together, the electrons orbiting each atom also get squeezed. This creates increasingly strong repulsive forces between electrons from neighboring atoms. Those repulsive forces are so intense that the electrons reorganize themselves into a new configuration, abandoning their normal orbitals and settling into the non-nuclear attractor sites instead.
It's almost like the electrons are being crowded out of their usual positions and forced to find alternative spots. The quantum mechanics work out so that electrons trapped in those non-nuclear sites have lower energy than if they tried to stay in their normal orbitals. Nature always favors lower energy configurations, so the electrons migrate to these interstitial positions and stay there.
What makes this phenomenon even more interesting is that electrides can form from multiple elements, not just sodium. Once scientists understood the basic mechanism, they began discovering and creating electrides from calcium, yttrium, lanthanum, and dozens of other elements. Some form only under extreme pressure like what exists in Earth's core. Others, remarkably, can now be synthesized right in a laboratory beaker at room temperature and atmospheric pressure.

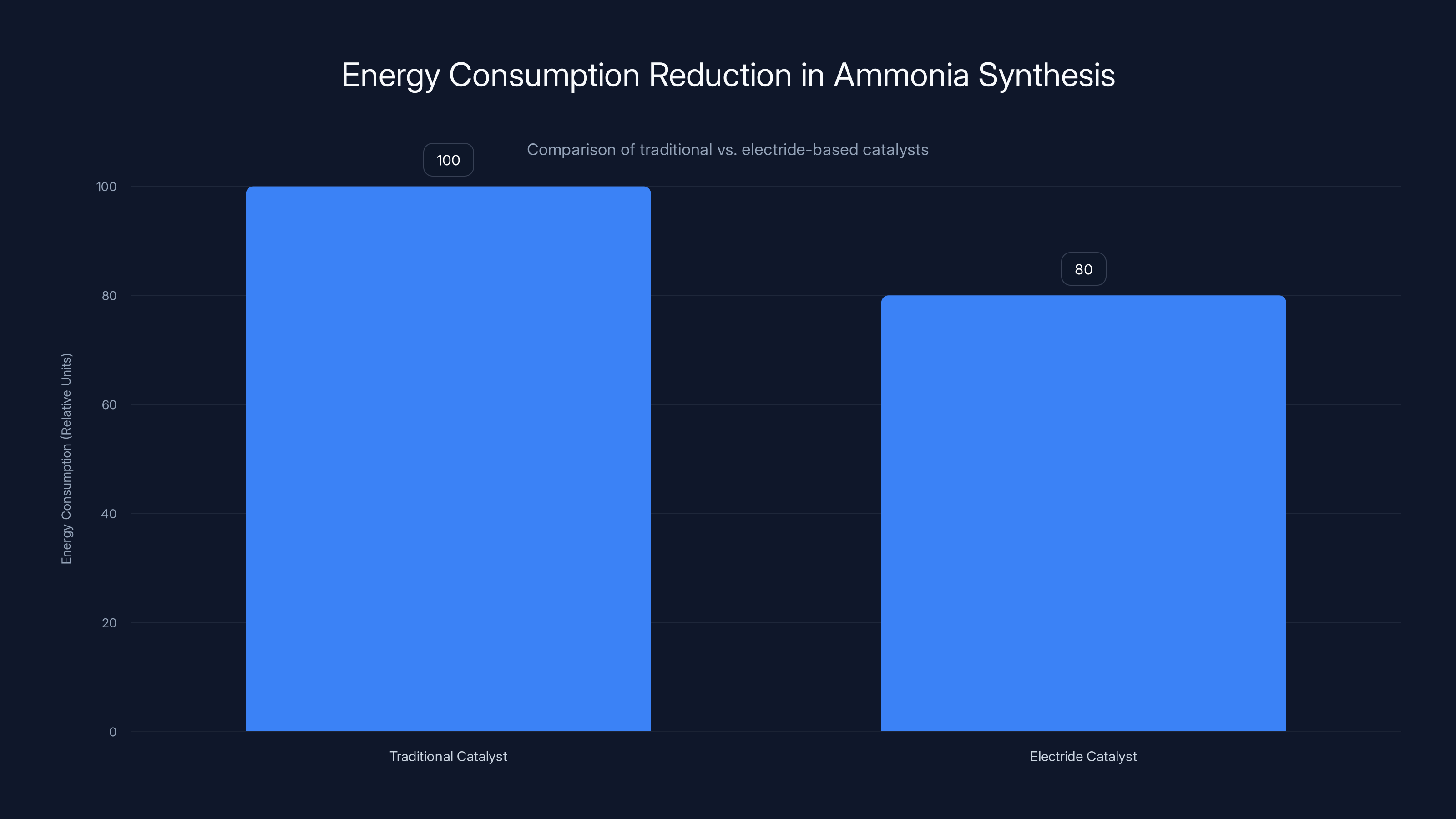
Electride-based catalysts reduce energy consumption in ammonia synthesis by 20% compared to traditional methods, marking a significant improvement in efficiency.
The Pressure Extreme: Earth's Inner Core Connection
To understand why electrides matter for solving Earth's elemental mystery, you need to grasp just how extreme conditions get at the center of our planet. We're not talking about the conditions in a laboratory, even a really well-equipped one. We're talking about pressures that exist nowhere else in our solar system except perhaps in the cores of gas giants.
Earth's inner core sits at the center of the planet, at depths around 1,220 kilometers below the surface. At that location, the pressure reaches something like 360 gigapascals. To put that number in perspective, you'd need to stack the weight of about 3.6 million Earth atmospheres on top of a single point to recreate that pressure. The deepest parts of the ocean, where you'd find those creepy anglerfish hanging out, have pressures of only about 100 megapascals. Earth's core is more than 3,000 times more extreme than that.
At these pressures, normal physics gets weird. Iron, which is what makes up Earth's core, behaves nothing like the iron in a steel beam or a cast iron skillet. The atoms get squeezed so tightly together that their electron clouds start overlapping and reorganizing, exactly like the theoretical predictions from the 1990s suggested.
According to research by Duck Young Kim and his colleagues at the Center for High Pressure Science & Technology Advanced Research in Shanghai, here's what might be happening. As the iron in Earth's core formed over the planet's history, lighter elements like hydrogen, carbon, nitrogen, and sulfur would have been present in the environment. Under the extreme pressures of the core, iron transforms into an electride form. This electride iron can absorb and trap these lighter elements at its non-nuclear attractor sites.
Think about it this way: imagine you have a sponge made of electride iron. The pores of this sponge are the non-nuclear attractor sites, perfect spots for light elements to settle into and get comfortable. Over billions of years, lighter elements would gradually diffuse into this spongy iron matrix, getting trapped in those electron-rich sites. The process might still be happening today, very slowly.
This hypothesis elegantly solves a major puzzle in geochemistry. Seismic waves traveling through Earth's inner core move at speeds that suggest the core is about 5 to 8 percent less dense than it would be if it were made of iron alone. That's a significant discrepancy. If the missing mass is because hydrogen, carbon, and nitrogen have infiltrated the iron and taken up space without adding much weight (since these elements are much lighter than iron), you'd get exactly the density deficit that seismic observations reveal.
The research team simulated this process in a computer. They modeled what happens to hydrogen atoms (standing in for other light elements) when they're surrounded by iron lattice at temperatures around 3,000 degrees Kelvin and pressures of 100 to 300 gigapascals. The simulations showed that at the higher pressures, hydrogen atoms would distribute themselves in a pattern that matches where electrons in an electride would sit. This distribution looked completely different from what you'd expect in normal metallic iron.
That pattern matching is compelling evidence that electride iron can indeed absorb hydrogen and probably other light elements too. If this process has been happening since Earth formed about 4.5 billion years ago, it could account for much of Earth's missing inventory of lighter elements.

How Sodium Electrides Revealed the Secret
Before researchers could propose that Earth's core contains electride iron absorbing lighter elements, they had to understand electrides themselves. That journey started with sodium, of all things, and a finding that shocked the geochemistry community.
Sodium is probably the most familiar element to most people, though they don't realize it. It's the sodium in table salt, in baking soda, in the sodium bicarbonate you use to clean your oven. In its pure metallic form, sodium is weird stuff. It's so soft you can cut it with a butter knife. It's so reactive with water that you shouldn't store it anywhere near moisture. Under normal conditions at Earth's surface, metallic sodium is shiny, reflective, and a decent conductor of electricity.
Then in 2009, researchers put sodium under 200 gigapascals of pressure and watched what happened. The element transformed. It stopped being a shiny metal. Instead, it became transparent like glass. More shocking still, it became an insulator instead of a conductor. This was completely unexpected by conventional theory.
Stephan Racioppi, a computational chemist who worked on understanding this phenomenon at the University of Buffalo, described the finding as very weird. Traditional quantum mechanical theories predicted that under such extreme pressure, sodium's electrons should become even more delocalized and free-flowing. The opposite happened. The theory was wrong.
What actually occurred was that sodium transformed into an electride. The extreme pressure forced its valence electrons into non-nuclear attractor sites. Those trapped electrons could no longer contribute to electrical conduction in the normal way. The material's optical properties changed too. Transparent electride sodium doesn't reflect light the way metallic sodium does because its electrons aren't moving through the lattice to interact with photons in the same way.
Once sodium electrides were discovered and understood, researchers began asking whether other elements could form electrides under pressure. The answer turned out to be yes. Calcium forms electrides. Yttrium forms electrides. Lanthanum does too. The list kept growing. Eventually, scientists realized that electrides might be more common in the universe than anyone had suspected.
The sodium discovery was crucial because it gave researchers a model system to study. They could conduct experiments on sodium electrides, measure their properties, run simulations, and build a theoretical framework for understanding electride chemistry. When they later hypothesized that iron in Earth's core might be an electride, they could draw on this foundation of sodium electride knowledge.
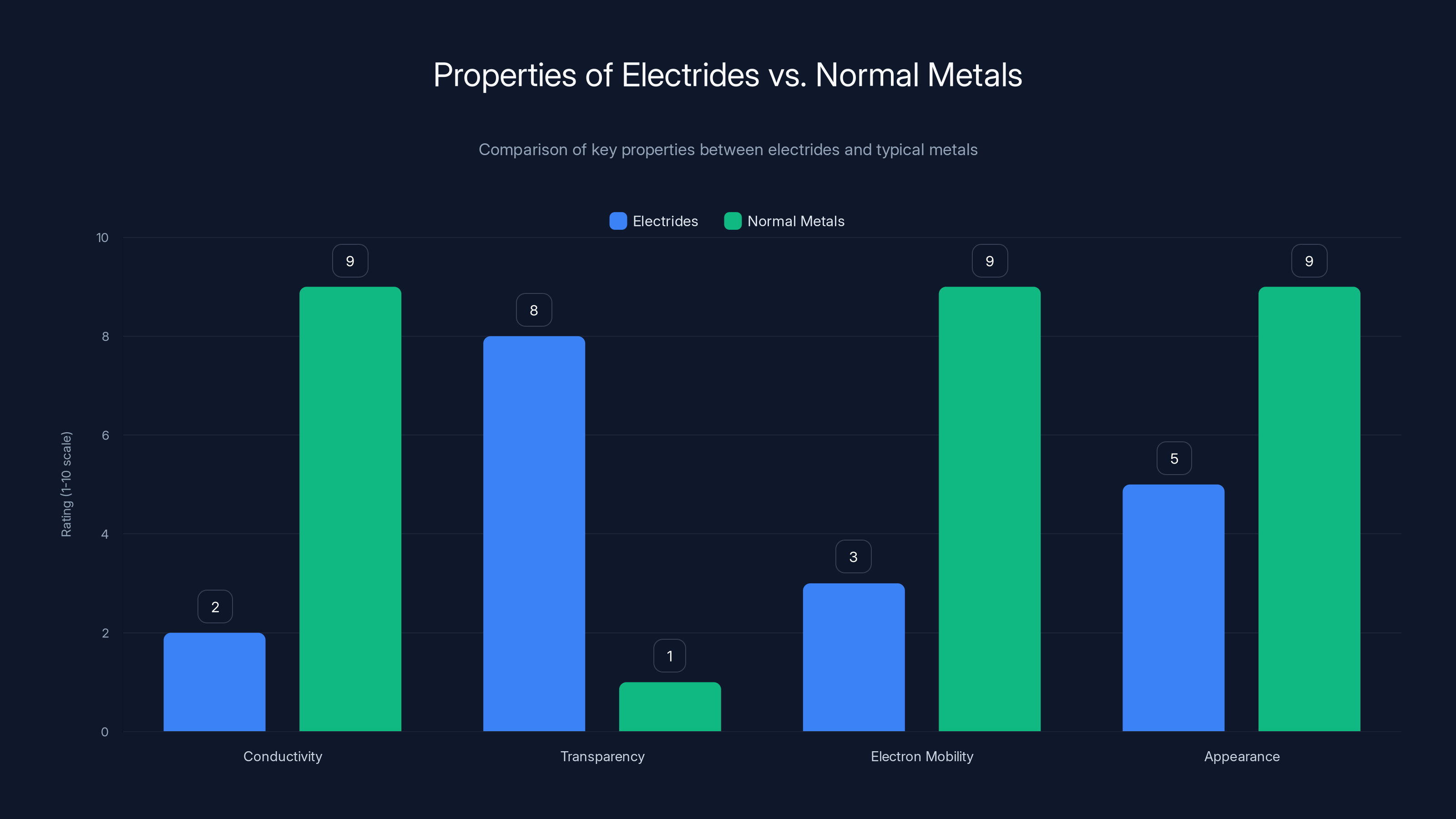
Electrides differ significantly from normal metals, showing low conductivity and high transparency, unlike typical metals which are excellent conductors and opaque. (Estimated data)
Room-Temperature Synthesis: From Extreme Pressures to Laboratory Benches
Here's where the story gets genuinely exciting for chemistry. For years, electrides were interesting only as theoretical predictions and high-pressure laboratory oddities. You needed massive equipment, extreme conditions, and sometimes months of preparation to create even tiny quantities of electride material. That severely limited their usefulness.
Then something remarkable happened. Chemists discovered they could make electrides at room temperature and normal atmospheric pressure. Not from high-pressure minerals, but from specially designed chemical precursors. This opened up an entirely new world of possibilities.
The breakthrough involved synthesizing electrides from layered crystal structures where atoms could be arranged in just the right geometric configuration to stabilize non-nuclear attractor sites even without extreme pressure. By carefully designing the crystal structure, chemists could create the electronic and spatial conditions that force electrons to occupy interstitial positions.
Imagine building a crystal where atoms are arranged like an apartment building with specific floor plans. If you design those floor plans carefully enough, you can create conditions where electrons would rather live in the hallways (non-nuclear attractors) than in any individual apartment (atomic orbitals). The electride forms spontaneously because it's the lowest energy state for the electrons, even without needing to squeeze the whole structure with massive pressure.
This was a game-changer. Suddenly, electrides became materials that chemists could synthesize, purify, and study with standard laboratory equipment. They could make larger quantities. They could test different compositions. They could explore the full range of what electrides could do.
One particularly successful approach involves using organic compounds to stabilize electride structures. Chemists create crystalline frameworks where inorganic atoms form the lattice, then incorporate organic molecules that help position electrons in non-nuclear sites. The result is a material with electride properties that's much easier to make and handle than high-pressure electrides.
The ability to create room-temperature electrides opened the door to practical applications. You couldn't realistically use an electride in an industrial process if you needed to maintain extreme pressure. But if you can make electrides in your chemistry lab and keep them stable at normal conditions, suddenly they become viable for real-world use.

The Catalysis Revolution: How Electrides Are Changing Chemistry
Now we're getting to the part that matters for humanity's future. Electrides have a special property that makes them extraordinarily useful as catalysts. A catalyst is a substance that speeds up a chemical reaction without being consumed in the process. Good catalysts are worth billions of dollars because they can make manufacturing processes faster, cheaper, and more efficient.
Electrides excel as catalysts because of their fundamental structure. Remember those non-nuclear attractor sites where electrons sit? Those sites are electron-rich. That means electrides have readily available electrons to donate to other molecules. Chemical reactions often happen because one molecule donates electrons to another molecule, or because electrons are transferred between molecules. Electrides, packed with electrons that are eager to move, are perfect for facilitating these electron-transfer reactions.
The first industrial application involves ammonia synthesis. Ammonia is one of the most important chemicals in the world. Roughly 80 percent of the nitrogen in fertilizer comes from synthesized ammonia. Without industrial ammonia production, modern agriculture literally couldn't exist. We couldn't feed 8 billion people without it.
Traditionally, ammonia is made using the Haber-Bosch process, which has been the dominant technology for over a century. It combines nitrogen gas from the air with hydrogen gas at high temperatures and pressures in the presence of an iron catalyst. The process works, but it's energy-intensive. Industrial ammonia plants account for about 1 to 2 percent of global energy consumption.
A Japanese team developed an electride-based catalyst for ammonia synthesis that reduces energy consumption by about 20 percent compared to traditional methods. That might sound modest, but for a global process that consumes as much energy as ammonia production does, a 20 percent improvement is enormous. It translates to billions of dollars in reduced energy costs and massive reductions in carbon emissions.
The electride catalyst works because it's better at facilitating the electron transfer needed to break apart the incredibly strong triple bond in nitrogen gas and combine those nitrogen atoms with hydrogen. The catalyst provides exactly the right electronic environment for this reaction to occur efficiently.

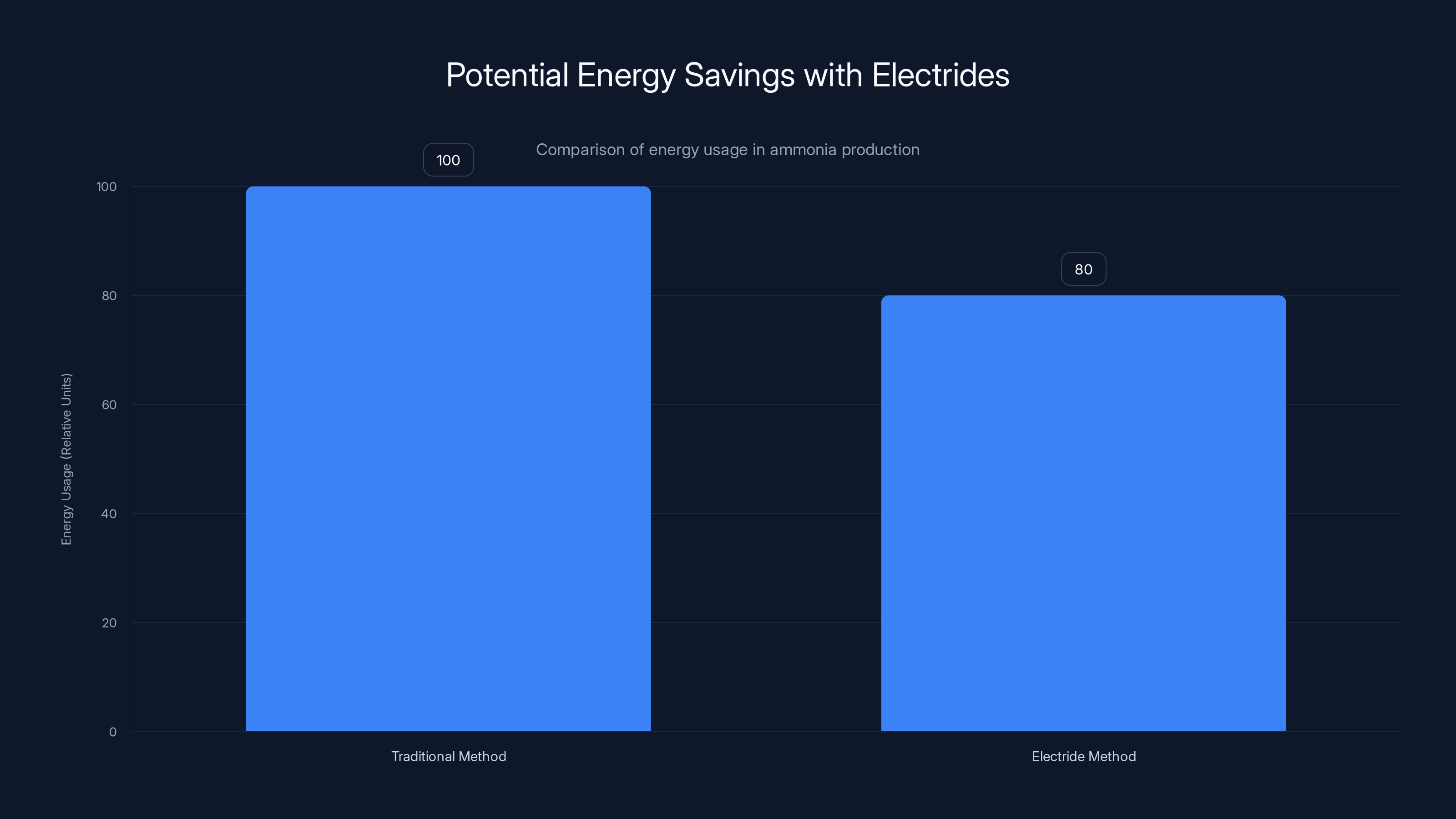
Electrides reduce energy consumption in ammonia production by approximately 20%, highlighting their potential for energy-efficient industrial applications. Estimated data.
Green Chemistry and Pharmaceutical Manufacturing
Ammonia production is just the beginning. Chemists are discovering that electrides can catalyze a wide range of reactions important for pharmaceutical manufacturing and fine chemical synthesis. These applications could be particularly transformative because pharmaceutical production involves many steps that currently rely on toxic catalysts or harsh reaction conditions.
One promising area involves carbon-carbon bond formation. Many pharmaceuticals need carbon atoms bonded together in very specific geometric arrangements. Creating these bonds efficiently requires good catalysts. Traditional catalysts for these reactions often involve palladium or other precious metals, which are expensive and sometimes involve toxic byproducts.
Electrides offer an alternative. Their electron-rich non-nuclear attractor sites can facilitate carbon bonding reactions in greener, cheaper ways. Early results show that electride catalysts can perform comparably to precious metal catalysts for certain reactions while using more abundant materials.
Another major pharmaceutical synthesis challenge involves oxidation reactions. Many drug syntheses require oxidizing specific parts of a molecule while leaving other parts untouched. This selectivity is difficult and often requires careful control of reaction conditions and catalysts. Electrides show promise for controlling these reactions more selectively, which could reduce waste and improve yields.
The beauty of using electrides for pharmaceutical synthesis is that they're less toxic than many traditional catalysts and can be synthesized from more abundant materials. Tungsten oxide electrides, for instance, are being studied because tungsten is more abundant and cheaper than palladium. If electride catalysts can perform comparably to precious metal catalysts, the entire pharmaceutical industry could shift toward cheaper, greener production methods.

The Theoretical Challenge: Understanding Electride Formation
For all the excitement about electride applications, chemists still face a fundamental challenge. We don't fully understand the chemical rules that govern when electrides form and when they don't. This is a crucial gap because filling it could help researchers design entirely new electrides with tailored properties.
The basic mechanism is clear enough. Under certain conditions (extreme pressure or specific crystal structure designs), electrons occupy non-nuclear attractor sites instead of atomic orbitals. But predicting which materials will form electrides, what pressure or conditions they need, and what properties the resulting electrides will have remains difficult.
This is where computational chemistry comes in. Researchers use quantum mechanics simulations to model electron behavior in different crystal structures and under different pressures. They calculate which arrangements have the lowest energy and therefore are most stable. These calculations can sometimes predict electride formation before it's observed experimentally.
But there's a catch. Quantum mechanical calculations are computationally expensive. For large crystal structures with many atoms, the calculations become impractical. Researchers need to make approximations, which introduces uncertainty. Sometimes the approximations work fine. Sometimes they lead predictions astray.
Stefano Racioppi and other computational chemists are working to develop better theoretical frameworks for predicting electride behavior. They're looking for patterns in the crystal structures that form electrides. They're investigating what properties of elements and their electron configurations make them more or less likely to form electrides.
One emerging pattern involves layered crystal structures. Many room-temperature electrides have layers of atoms with larger gaps between the layers. These gaps seem to be where non-nuclear attractor sites naturally form. Understanding why layered structures favor electride formation could guide the design of new electrides.
Another pattern involves the number of electrons relative to the number of atoms. Some electrides seem to form when there are more electrons than the atoms need to form normal chemical bonds. The excess electrons then settle into non-nuclear attractor sites rather than staying in atomic orbitals. If this pattern holds generally, it could be a design principle for creating new electrides.

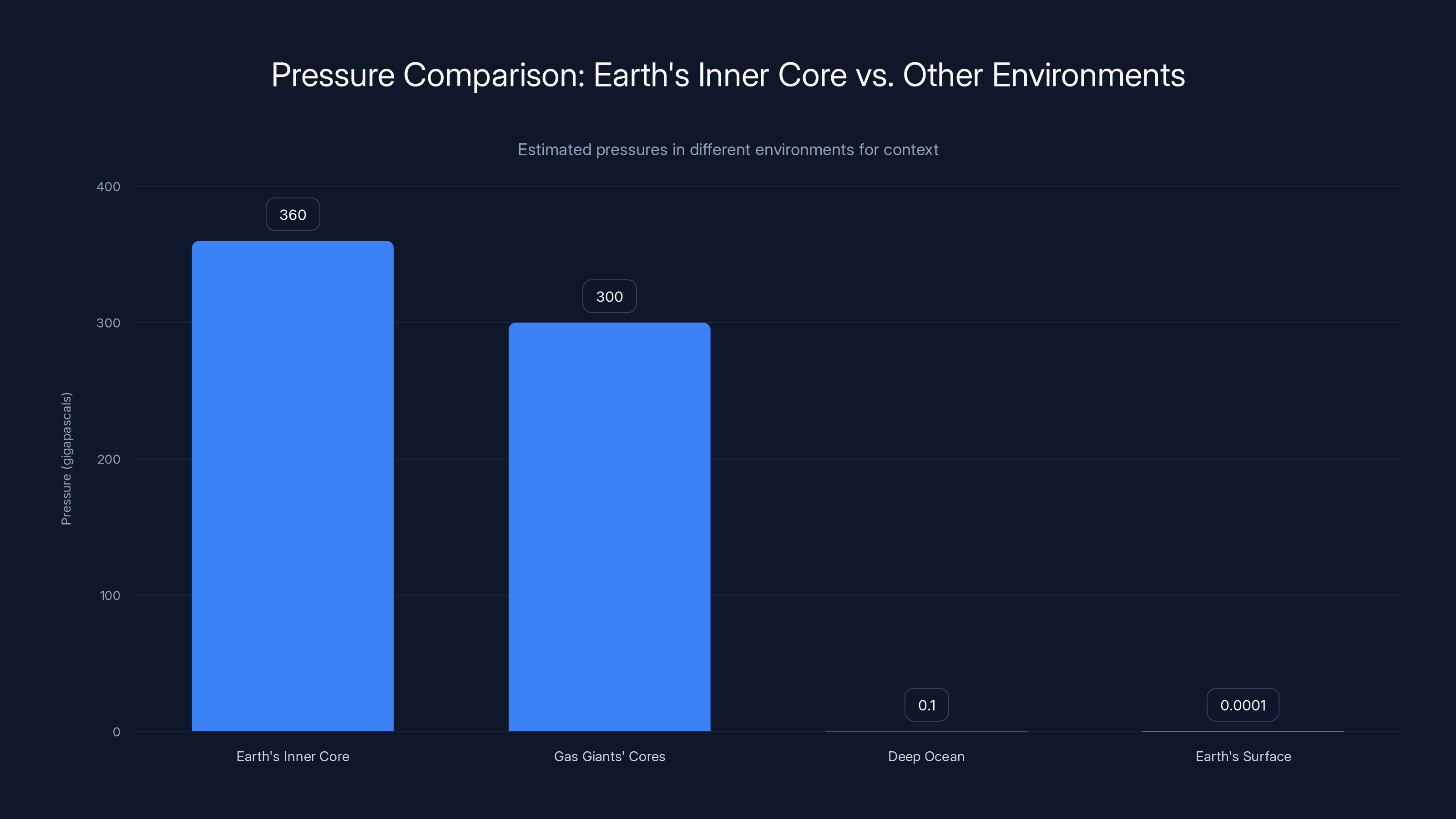
Earth's inner core experiences pressures of around 360 gigapascals, vastly exceeding those found in other environments, such as gas giants' cores and the deep ocean. Estimated data.
The Earth Science Connection: Solving the Planetary Mystery
Circling back to where we started, the electride explanation for Earth's missing elements represents a potential breakthrough in understanding planetary chemistry. For decades, geoscientists could measure that Earth had significantly less hydrogen, carbon, nitrogen, sulfur, and noble gases than the Sun and certain meteorites. But they couldn't explain where these elements went.
Various hypotheses existed. Some elements escaped to space early in Earth's history, that's true. But that couldn't account for the full deficit. Some suggested that these elements were locked in Earth's core, but they couldn't explain the mechanism.
The electride hypothesis provides a mechanism. It explains how Earth's core, which formed extremely hot and would normally have excluded these lighter elements, could somehow incorporate and retain them. The answer: once the core cooled enough for iron to crystallize into solid form, and given the extreme pressure conditions, iron became an electride. Electride iron can absorb and trap lighter elements that would normally be excluded from metallic iron.
Duck Young Kim's team estimated that this absorption process might occur gradually over billions of years. Elements would slowly diffuse into the electride iron from the surrounding material, getting trapped in non-nuclear attractor sites. This process could theoretically still be happening today, though extremely slowly given the vast distances involved and the extremely low temperatures (on a atomic scale) where diffusion occurs.
The evidence supporting this hypothesis is intriguing. The seismic wave velocity measurements showing the core is 5 to 8 percent less dense than pure iron match what you'd expect if lighter elements had infiltrated the iron. The computer simulations showing hydrogen atoms arranging themselves in the pattern of electride non-nuclear attractor sites provide experimental support for the mechanism.
But this remains a hypothesis that needs further testing. Researchers need to study electride iron under core conditions more extensively. They need to measure how quickly lighter elements actually diffuse into electride iron. They need to understand whether the non-nuclear attractor sites in electride iron can remain stable over geological timescales.

Advanced Electride Variants and Future Possibilities
Beyond sodium and the room-temperature electrides already being used commercially, chemists are exploring electrides made from an astonishing variety of elements. Calcium, magnesium, strontium, barium, lanthanum, yttrium, and dozens of other elements have shown electride character under appropriate conditions.
Each different element produces electrides with different properties. Calcium electrides behave differently from sodium electrides. The catalytic properties vary. The stability under different conditions changes. This diversity is actually good news for materials scientists because it means there's likely an electride optimized for nearly any application you can imagine.
Researchers are discovering electrides with remarkable properties. Some show enhanced conductivity at room temperature. Others demonstrate specific reactivity that could be valuable for certain chemical transformations. A few even show magnetic properties that weren't predicted but might be useful for materials applications.
One particularly exciting development involves organic electrides, where organic molecules play a role in the crystal structure. These hybrid organic-inorganic materials can be synthesized in creative ways, using organic chemistry techniques alongside traditional solid-state chemistry. This opens possibilities for making electrides with properties that purely inorganic materials can't achieve.
Another frontier involves electrides in nanoscale form. Instead of bulk crystals, researchers are learning to synthesize electride particles with dimensions measured in billionths of a meter. At these tiny scales, the surfaces have different properties than bulk electrides, potentially offering even greater catalytic activity because more atoms are at the surface where reactions happen.

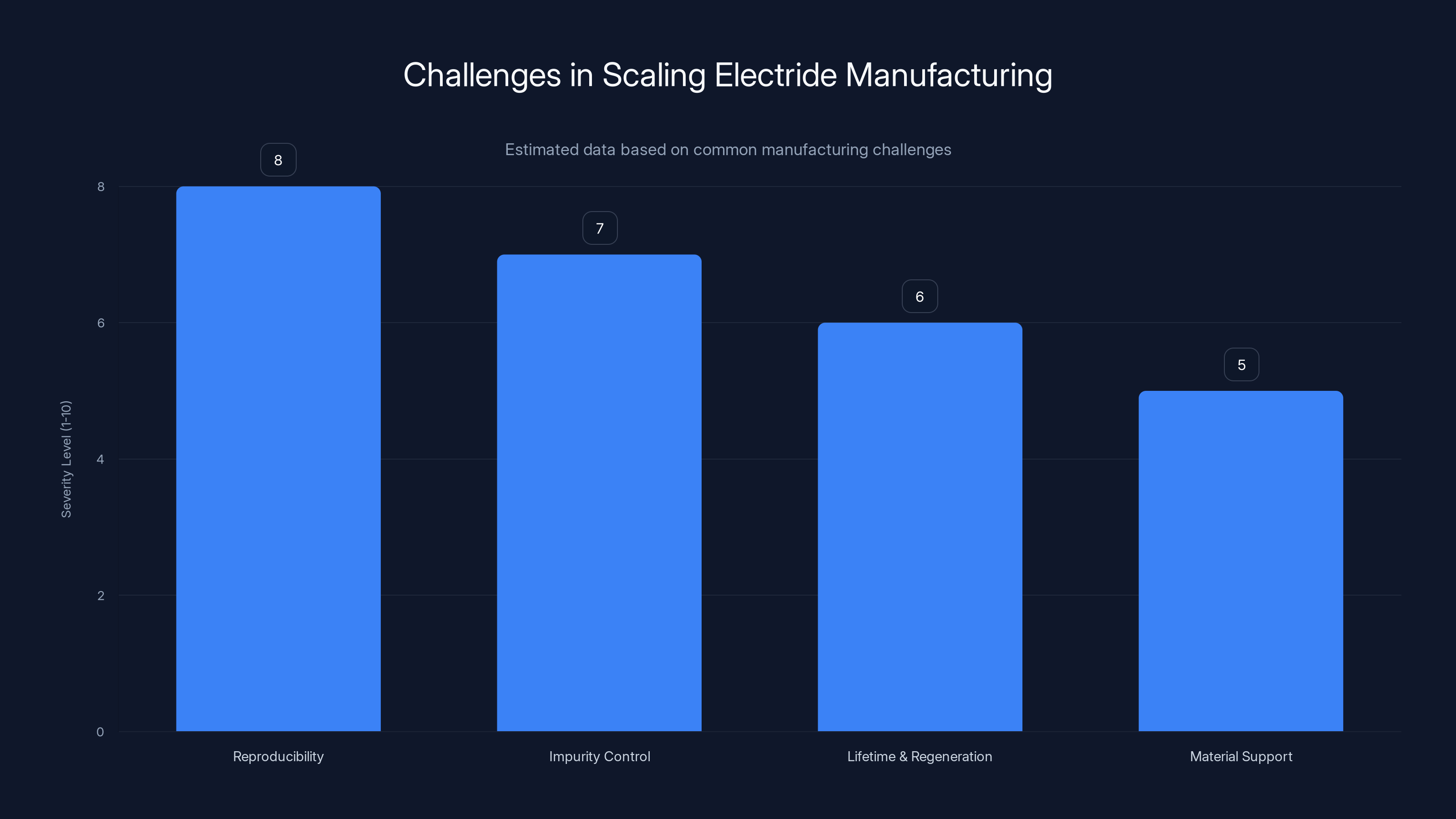
Reproducibility and impurity control are major challenges in scaling electride manufacturing, with severity levels estimated at 8 and 7 respectively. Estimated data.
Manufacturing Challenges and Scalability
Despite the exciting laboratory discoveries and initial commercial applications, bringing electrides to mass production still presents significant challenges. The Japanese ammonia synthesis process using electride catalysts works beautifully in demonstration plants, but scaling it up to replace traditional ammonia production worldwide is a different proposition entirely.
One challenge is reproducibility. Making small quantities of high-quality electride material in a research lab is one thing. Manufacturing consistent electride catalysts in industrial quantities is quite another. Tiny variations in synthesis conditions can dramatically affect the final product's properties. Getting the synthesis process down to a reproducible, cost-effective procedure suitable for large-scale production requires substantial development.
Another challenge involves understanding and controlling impurities. In the laboratory, chemists work under carefully controlled conditions with ultra-pure starting materials. In an industrial setting, these materials are more expensive and harder to maintain in pure form. Impurities in the electride can drastically reduce catalytic activity or cause unwanted side reactions.
Then there's the matter of lifetime and regeneration. Catalysts get used up over time. They lose activity as the material decomposes or becomes blocked with products. A commercial process needs to understand how long an electride catalyst lasts before needing replacement or regeneration, and whether it can be regenerated efficiently.
The ammonia synthesis electride catalyst appears to have solved many of these challenges, which is why it's already entering commercial use. But for other electride applications being researched—pharmaceutical synthesis, specialty chemicals, fine organic synthesis—the path to commercial viability remains less clear.
Researchers are also exploring whether electrides can be supported on other materials, like metal oxides, to improve their stability and reusability. A catalyst supported on an inert material might be more robust and easier to recover and regenerate than a free-standing electride particle.

Environmental and Sustainability Implications
If electrides can live up to their promise in catalysis, the environmental implications could be substantial. Consider ammonia production alone. The traditional Haber-Bosch process consumes roughly 1 to 2 percent of global energy production and generates significant carbon emissions. If electride catalysts could be deployed worldwide with even modest efficiency improvements, we're talking about saving gigatons of carbon dioxide annually.
Expand that to all the other chemical manufacturing processes that could benefit from electride catalysts. Pharmaceutical production is notoriously wasteful, generating tens of thousands of tons of byproducts for each ton of useful drug. If electride catalysts could improve reaction selectivity and reduce waste, that's a huge environmental win.
Beyond the direct environmental benefits of more efficient catalysis, there's a more subtle benefit. Electrides can be made from relatively abundant elements. Many promising electrides use calcium, magnesium, tungsten, or other elements that are far more abundant than precious metals like platinum, palladium, and rhodium that traditional catalysts rely on. This means electride catalysis has the potential to be more sustainable long-term because it doesn't depend on mining rare and precious metals.
Of course, sustainability depends on how you source the precursor materials and what you do with spent catalysts. A truly sustainable electride catalyst industry would need to develop good recycling and regeneration practices, use renewable energy in synthesis, and source raw materials responsibly. But the potential is there.

Interdisciplinary Research and Future Directions
What's remarkable about electride research is how it bridges multiple scientific disciplines. You need geochemistry to understand Earth's core composition. You need solid-state physics to explain electron behavior at extreme pressures. You need quantum chemistry to predict which materials form electrides. You need materials science to synthesize them. You need chemical engineering to scale them up. You need industrial chemistry to apply them to real production processes.
This interdisciplinary nature means that progress in any one area tends to accelerate progress in others. When geochemists propose that electride iron might exist in Earth's core, it motivates physicists and chemists to study iron electrides more carefully. When chemists discover new electride materials, it opens possibilities for new catalytic applications that engineers then explore. When applications emerge, it creates funding and motivation for more fundamental research.
The coming years will likely see accelerated discovery of new electrides. Machine learning is being applied to predict which crystal structures might form electrides without needing to calculate quantum mechanics by hand for every possibility. High-throughput screening techniques can test many potential electride materials simultaneously. Databases of computational results are being built to guide new experiments.
We'll probably see electrides expanding beyond catalysis too. Some electrides show interesting electronic and optical properties that might make them useful for semiconductors, energy storage, or other materials applications. The ability to trap electrons in non-nuclear sites opens up possibilities that traditional materials don't offer.
There's also the tantalizing possibility of discovering electrides in nature beyond Earth's core. Some meteorites might contain electride phases. Planets with extreme internal pressures might have thick layers of electride material. Understanding natural electrides could tell us about planetary formation throughout the universe.

FAQ
What exactly is an electride and how is it different from normal metals?
An electride is a solid material where electrons occupy specific sites between atoms (called non-nuclear attractors) instead of orbiting individual atomic nuclei as they do in normal metals. While metals are typically good electrical conductors because electrons flow freely through them, electrides lose this property because their trapped electrons can't move freely. This can transform a shiny, reflective metal like sodium into a transparent, insulating material at high pressure.
How do electrides form, and why is extreme pressure necessary for some but not others?
Electrides form when electrons experience conditions that make non-nuclear attractor sites lower in energy than normal atomic orbitals. At extreme pressures, atoms get squeezed so closely together that electrons from neighboring atoms repel each other intensely, forcing electrons to relocate to non-nuclear sites. However, scientists have learned to design special crystal structures that create these electron-trapping conditions even at room temperature and normal pressure by carefully arranging atoms in layered or specific geometric patterns.
Why might Earth's missing lighter elements be hidden in the core as an electride?
Under the extreme pressure of Earth's core (360 gigapascals), iron transforms into an electride form capable of trapping and absorbing lighter elements like hydrogen, carbon, nitrogen, and sulfur. Computer simulations show that these lighter elements would naturally migrate to non-nuclear attractor sites in electride iron. Over billions of years, this gradual absorption process could explain where Earth's missing inventory of lighter elements went and why seismic data indicates the core is 5 to 8 percent less dense than pure iron would be.
How are electrides being used commercially right now?
The most developed commercial application is in ammonia synthesis, where Japanese researchers have deployed an electride catalyst that reduces energy consumption by approximately 20 percent compared to the traditional Haber-Bosch process. Ammonia production accounts for a substantial portion of global industrial energy use, so this improvement translates to billions of dollars in energy savings and significant carbon emission reductions. Researchers are actively developing electride catalysts for pharmaceutical manufacturing and other green chemistry applications.
What are non-nuclear attractors and why do electrons stay trapped there?
Non-nuclear attractors are geometrically specific locations in a crystal structure where electrons can occupy stable positions despite the absence of an atomic nucleus to anchor them directly. Electrons are held in place by the electrostatic attraction of surrounding atomic nuclei and the quantum mechanical requirement that electrons occupy the lowest available energy states. Once electrons settle into these sites at non-nuclear attractors, they remain trapped because moving away from these positions would increase their energy.
What challenges remain before electrides become widely used in industry?
The primary challenges include developing reproducible, cost-effective synthesis methods for large-scale manufacturing, understanding how impurities affect electride properties, determining how long electride catalysts remain active before needing replacement, and developing regeneration or recycling methods for spent catalysts. Additionally, researchers need to discover more electride materials with tailored properties for specific applications and develop better theoretical frameworks for predicting which materials will form electrides.
Could electrides replace precious metal catalysts like platinum and palladium?
Electrides show significant promise for replacing precious metal catalysts in certain applications because they can be synthesized from more abundant elements and often demonstrate comparable or superior catalytic performance. However, this transition won't happen across the board. Some reactions work better with precious metal catalysts, and the properties needed depend heavily on the specific chemical transformation being catalyzed. The realistic expectation is that electrides will capture significant market share from precious metals in applications like ammonia synthesis and certain pharmaceutical syntheses.
How does the discovery of electrides change our understanding of Earth's composition?
The electride hypothesis provides a mechanism for Earth's core to chemically incorporate and retain lighter elements that would normally be excluded from metallic iron. This potentially solves a century-old mystery about why Earth is depleted in hydrogen, carbon, nitrogen, sulfur, and noble gases compared to the Sun and some meteorites. If confirmed, this discovery would fundamentally change how scientists model Earth's interior and understand our planet's formation and chemical evolution.
Are there naturally occurring electrides besides Earth's core?
While electride iron in Earth's core remains theoretical, scientists have speculated that electrides might exist in meteorites and potentially in the interiors of other planets. Some meteorites contain materials that formed under extreme pressure and might include electride phases. Planets with thick, high-pressure interiors might have extensive electride layers. Discovering natural electrides beyond Earth would provide additional confirmation of electride stability and potentially reveal new electride compositions not yet synthesized in laboratories.
What's the difference between inorganic electrides and organic-inorganic hybrid electrides?
Inorganic electrides are made entirely from inorganic atoms arranged in crystal structures that stabilize non-nuclear attractor sites. Organic-inorganic hybrid electrides combine inorganic crystal lattices with organic molecules that help position and stabilize electrons in non-nuclear sites. Hybrid electrides often have the advantage of being more easily synthesized at room temperature without extreme pressure, and the organic components can be modified to fine-tune the material's properties for specific applications.

The Future of Materials Science and Planetary Understanding
Electrides represent one of those rare moments in science when a discovery that seemed merely theoretical suddenly becomes relevant to both fundamental planetary science and practical engineering. The fact that the same unusual material might explain where Earth's missing elements went while also revolutionizing how we make fertilizer and pharmaceuticals speaks to the deep interconnectedness of chemistry.
The research ahead is exciting. Geochemists will continue testing whether electride iron at Earth's core can actually absorb lighter elements as quickly as the planetary mystery requires. They'll develop better techniques for studying materials at core pressures and temperatures. Spectroscopy studies might detect signatures of light elements in core samples, if we ever get samples from the core (which we can't currently do directly, but we might infer from seismic data or meteorite composition).
Chemists will continue discovering new electrides and finding new applications for them. The most near-term opportunities probably involve replacing precious metal catalysts in pharmaceutical synthesis and fine chemicals manufacturing. Medium-term opportunities might include electride-based batteries or other energy storage materials. Long-term possibilities include materials with properties we can barely imagine right now.
Engineers will work on scaling up electride synthesis and learning how to produce them reliably at industrial scale. As manufacturing improves and costs drop, electride catalysts will become economical for more and more applications. The efficiency gains will compound. The environmental benefits will multiply.
The story of electrides shows how science progresses. A century-old puzzle about Earth's composition pushes researchers to propose an exotic solution. That solution motivates fundamental research into an unusual form of matter. Understanding that matter opens up practical applications. Those applications create incentives for more research, which leads to new discoveries and further applications. It's a virtuous cycle, and we're only at the beginning of it with electrides.
For anyone working in chemistry, materials science, or geochemistry, electrides are a material to watch closely. They're not just academic curiosities. They're real materials that could reshape major industrial processes and help us build a more sustainable future while simultaneously solving mysteries about our own planet that have puzzled scientists for generations. That's the kind of convergence where real breakthroughs happen.
Key Takeaways
- Electrides are materials where electrons occupy non-nuclear attractor sites rather than atomic orbitals, giving them exotic properties completely different from normal metals.
- Earth's missing lighter elements (hydrogen, carbon, nitrogen) may be trapped in electride iron deep in the core at 360 gigapascals of pressure, explaining a 5-8% density deficit detected by seismic data.
- Room-temperature electride synthesis breakthroughs have enabled practical applications in catalysis, particularly a 20% energy reduction in industrial ammonia production.
- Electride catalysts show promise for replacing precious metal catalysts in pharmaceutical and fine chemical synthesis, offering more sustainable and economical alternatives.
- Discovering and understanding the rules governing electride formation remains the key challenge for expanding their use across industrial chemistry.
![Electrides: The Mysterious Materials Hiding Earth's Missing Elements [2025]](https://tryrunable.com/blog/electrides-the-mysterious-materials-hiding-earth-s-missing-e/image-1-1769526521412.jpg)



