Introduction
Europe has taken a significant step forward by authorizing Moderna's mRNA-based combination vaccine, designed to combat both influenza and COVID-19. This marks a pivotal moment in the global healthcare landscape, as Europe becomes the first region to approve such a dual-purpose vaccine. Meanwhile, the U.S., despite being the development ground for this vaccine, has yet to authorize it. This article delves into the scientific, regulatory, and societal implications of this approval.
TL; DR
- Europe leads with the first mRNA combo vaccine for flu and COVID-19, while the U.S. holds back. According to Reuters, this approval sets a precedent for other regions.
- Moderna's mRNA-1083 vaccine simplifies immunization, targeting two major respiratory viruses in one shot. Fierce Pharma highlights the vaccine's potential to streamline vaccination efforts.
- The vaccine's approval process in Europe highlights differences in regulatory approaches compared to the U.S. The Ars Technica article discusses these regulatory differences.
- Potential global impact on vaccination strategies as more countries consider dual-purpose vaccines. Johns Hopkins University explores the broader implications for public health.
- Future trends include increased investment in multi-target vaccines and potential expansion to other viruses. IndexBox forecasts growth in the nasal vaccines market, driven by preferences for needle-free options.
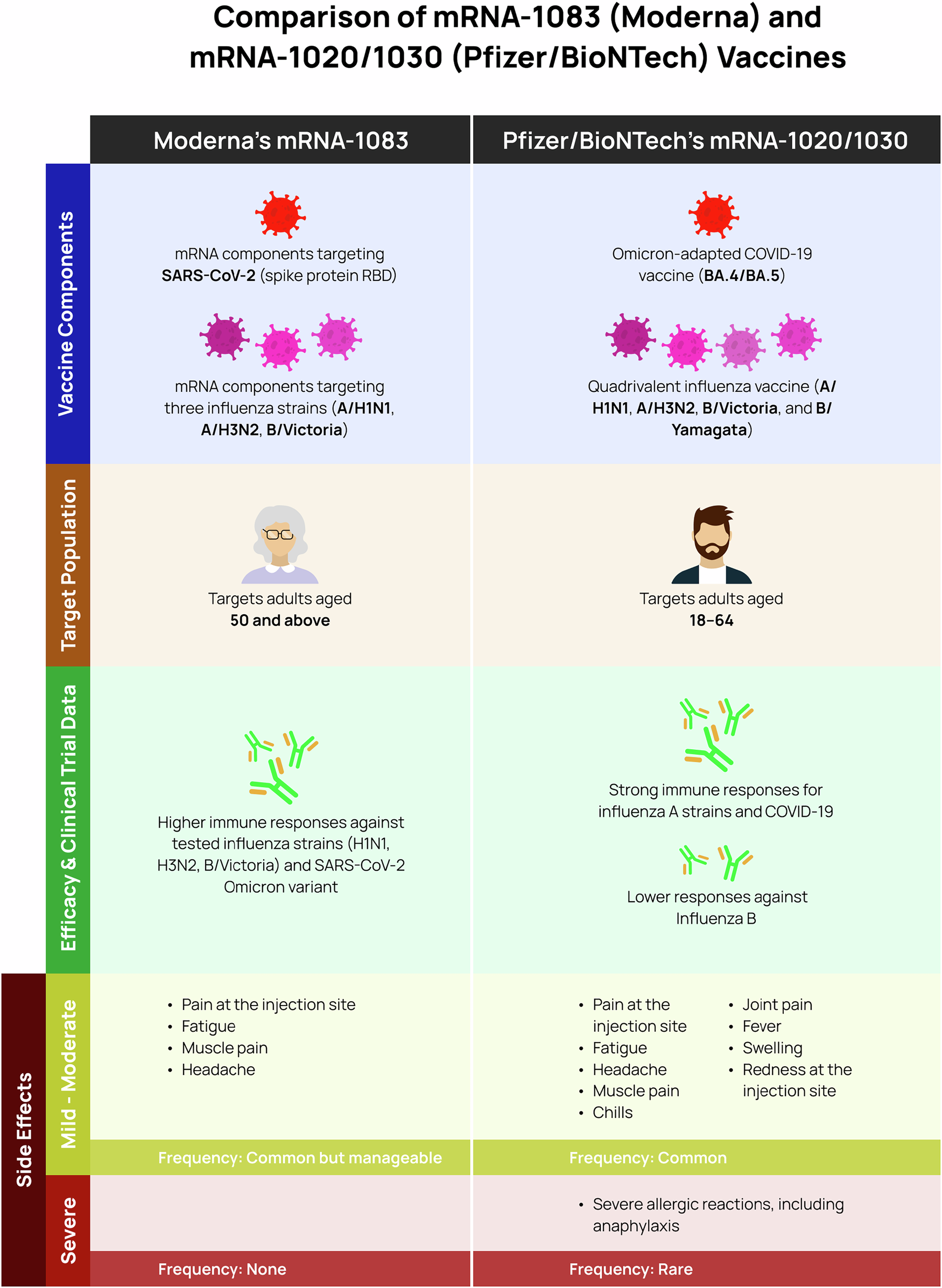
The clinical trials for mRNA-1083 progressed from small-scale safety trials to large-scale efficacy confirmation, involving an estimated 3,600 participants in total. Estimated data.
The Science Behind mRNA Vaccines
To understand the significance of Moderna's mRNA-1083, it's essential to grasp the basics of mRNA technology. mRNA vaccines work by introducing a small piece of genetic material from the virus into the body. This material instructs cells to produce a harmless spike protein, which triggers an immune response without causing the disease. Britannica provides a comprehensive overview of how mRNA vaccines function.
How mRNA Vaccines Work
- Injection: The vaccine is injected into the muscle, where cells take up the mRNA.
- Protein Production: Cells use the mRNA instructions to produce the virus's spike protein.
- Immune Response: The immune system recognizes the spike protein as foreign and creates antibodies.
- Protection: If exposed to the actual virus later, the immune system can quickly respond.
Advantages of mRNA Technology
- Rapid Development: mRNA vaccines can be designed quickly once the genetic sequence of the virus is known. The Memorial Sloan Kettering Cancer Center highlights the rapid development capabilities of mRNA technology.
- High Efficacy: Clinical trials have shown mRNA vaccines to be highly effective in preventing diseases. Clinical Trials Arena reports on the high efficacy rates observed in trials.
- Adaptability: mRNA technology can be easily adjusted to target new virus strains.

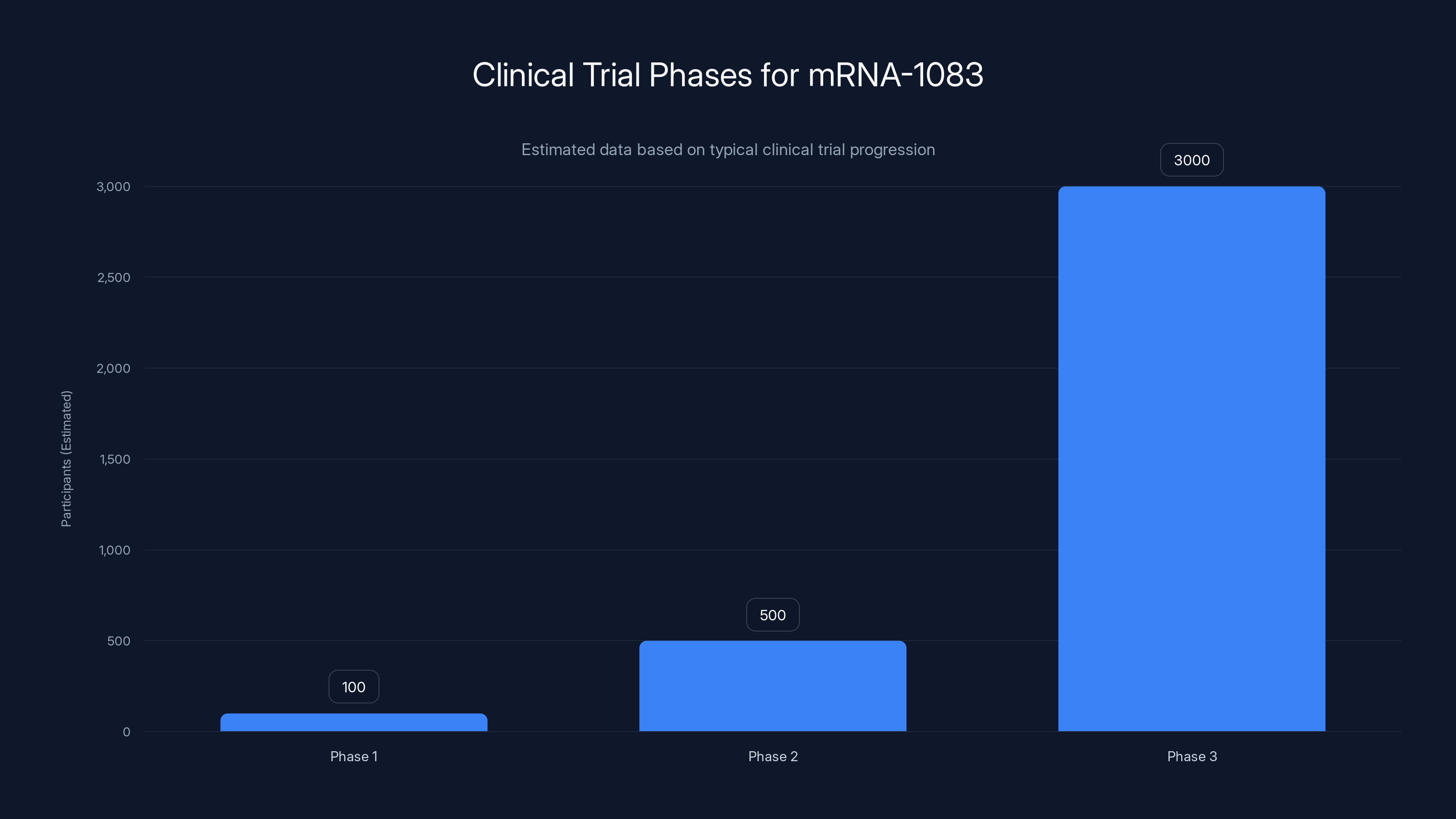
The mRNA-1083 combo vaccine is expected to increase vaccination rates by simplifying the immunization process and reducing healthcare costs. (Estimated data)
The Road to Approval in Europe
The European Medicines Agency (EMA) plays a critical role in the vaccine approval process. For mRNA-1083, the journey began with extensive clinical trials. Stock Titan details the EMA's approval process for Moderna's vaccine.
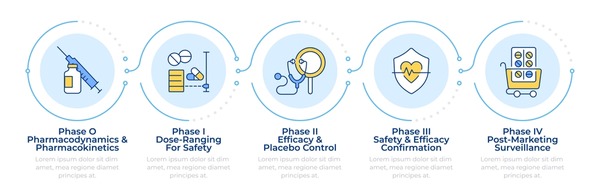
Clinical Trials Overview
- Phase 1: Initial safety trials on a small group to determine the appropriate dosage.
- Phase 2: Expanded trials to evaluate efficacy and side effects.
- Phase 3: Large-scale trials to confirm efficacy and monitor adverse reactions.
Key Findings
- High Efficacy: mRNA-1083 showed efficacy rates above 90% against both flu and COVID-19. Reuters reports on the vaccine's high efficacy.
- Safety: The vaccine demonstrated a robust safety profile, with side effects similar to those of existing vaccines.
- Convenience: A single dose simplifies logistics and increases vaccination rates.
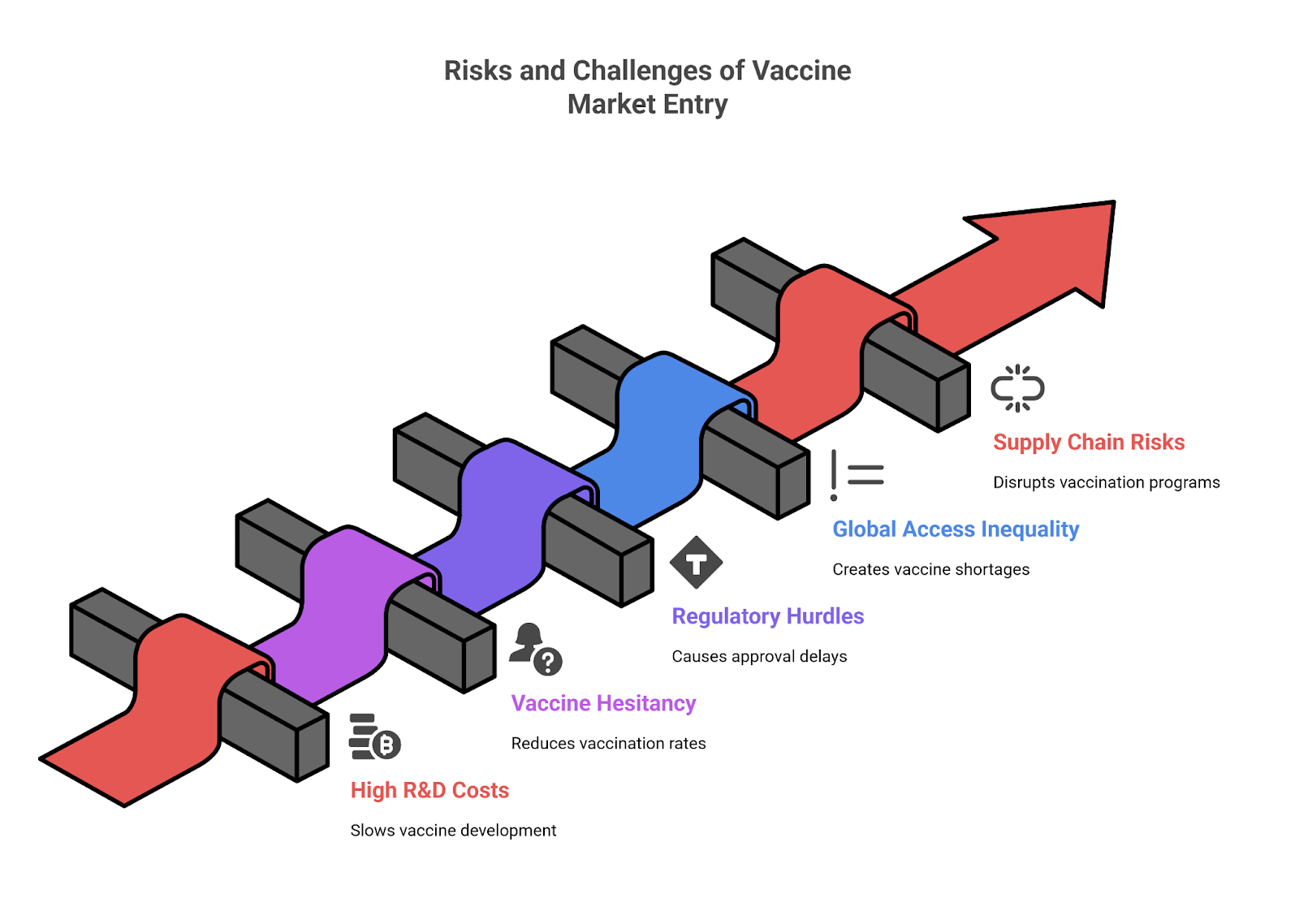
Why the U.S. Lags Behind
Despite its development in the U.S., the combination vaccine has not yet received FDA approval. Several factors contribute to this delay.
Regulatory Hurdles
The FDA requires thorough evaluation of combination vaccines to ensure safety and efficacy. The U.S. regulatory framework is more cautious, often requiring additional data. Clinical Trials Arena discusses the stringent requirements of the FDA.
Political and Societal Factors
- Vaccine Hesitancy: Political movements and misinformation have fueled skepticism about new vaccines. WATTAgNet highlights the impact of vaccine hesitancy on public health initiatives.
- Public Health Priorities: Focus on single-virus vaccines and boosters has delayed combo vaccine consideration.
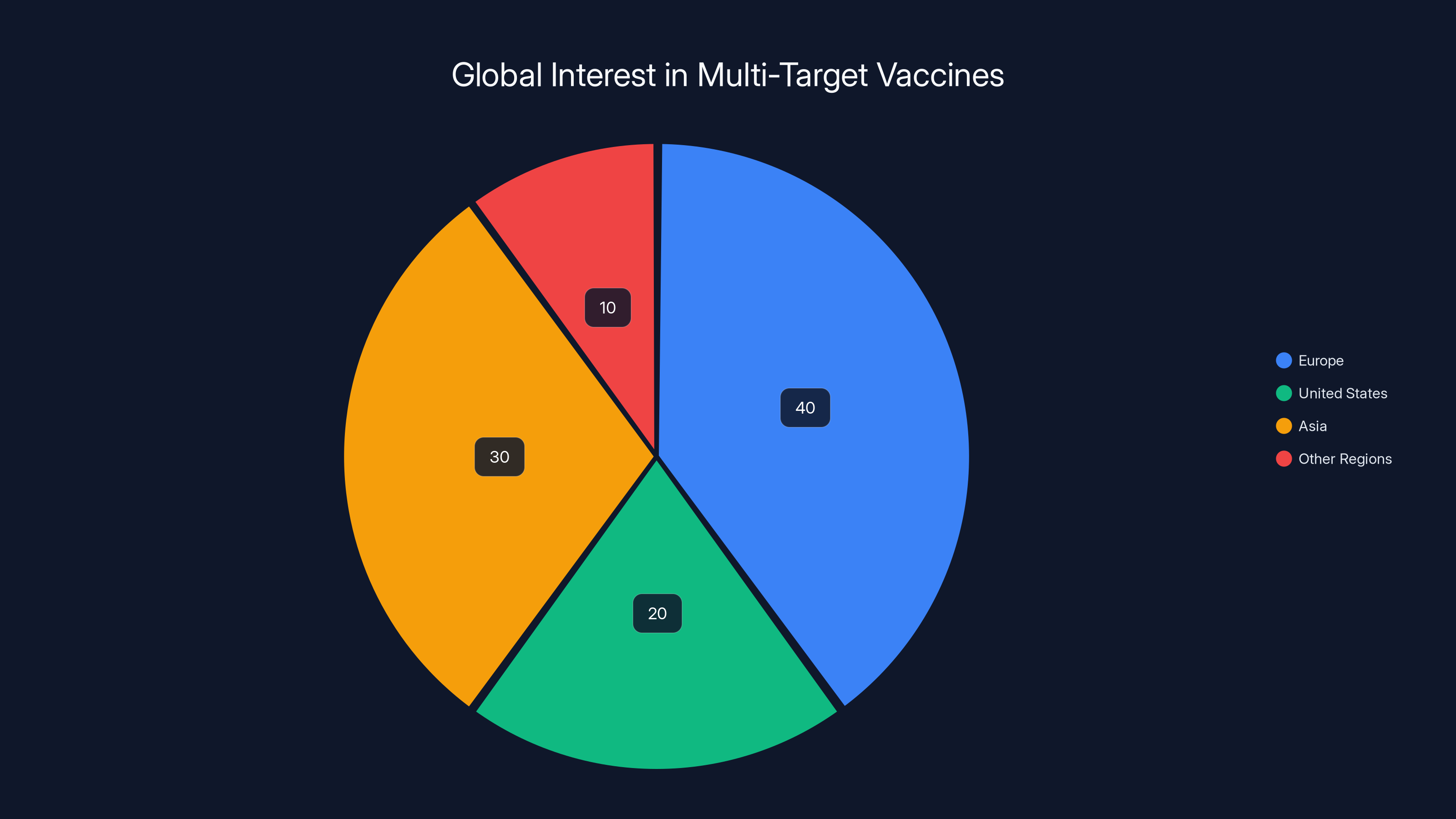
Europe shows the highest interest in multi-target vaccines, driven by regulatory approval of the mRNA combo vaccine. Estimated data.
Global Implications and Future Trends
Europe's approval of the mRNA-1083 vaccine sets a precedent for other regions. It could lead to a shift in global vaccination strategies.
Potential Impact
- Increased Vaccine Uptake: Simplified immunization could boost vaccination rates. IndexBox discusses how simplified vaccine delivery methods can increase uptake.
- Cost-Effectiveness: A single vaccine dose reduces logistical expenses and healthcare costs.
- Broader Protection: Dual protection against flu and COVID-19 could reduce the burden on healthcare systems.
Future Trends
- Expansion to Other Viruses: Researchers are exploring mRNA vaccines targeting multiple pathogens. Memorial Sloan Kettering Cancer Center is investigating the potential of mRNA vaccines for other diseases.
- Innovation in Vaccine Delivery: New methods, such as nasal sprays, are being developed to improve accessibility.
- Focus on High-Risk Populations: Targeted vaccination strategies for vulnerable groups could further improve outcomes.
Practical Implementation: What It Means for Healthcare Providers
Healthcare providers in Europe must now adapt to the introduction of the mRNA-1083 vaccine.
Implementation Strategies
- Training: Educate healthcare workers on the new vaccine's specifics and administration techniques.
- Storage and Handling: Ensure proper storage facilities to maintain vaccine efficacy.
- Patient Education: Inform patients about the benefits and safety of the combination vaccine.
Challenges and Solutions
While the introduction of mRNA-1083 is promising, it comes with challenges.
Common Pitfalls
- Logistical Issues: Coordinating distribution and administration across diverse healthcare settings.
- Public Skepticism: Overcoming misinformation and vaccine hesitancy.
Solutions
- Clear Communication: Transparent communication strategies to build public trust. Reuters emphasizes the importance of clear communication in overcoming public skepticism.
- Collaboration: Partnerships with local organizations to facilitate vaccine rollout.

Conclusion: A New Era in Vaccination
The approval of Moderna's mRNA-1083 in Europe marks the beginning of a new era in vaccination. By combining protection against two significant viruses, this vaccine represents a major advancement in healthcare. As other regions observe Europe's progress, the adoption of combo vaccines could become a global trend, reshaping vaccination strategies worldwide.

FAQ
What is mRNA-1083?
mRNA-1083 is a combination mRNA vaccine developed by Moderna to provide protection against both influenza and COVID-19 in a single shot.
How does the vaccine work?
The vaccine uses mRNA technology to instruct cells to produce spike proteins from both viruses, prompting an immune response.
Why was it approved in Europe first?
Europe's regulatory body, the EMA, expedited the approval process based on positive trial results, while the U.S. FDA has been more cautious.
What are the benefits of a combo vaccine?
It simplifies immunization by reducing the number of shots needed, potentially increasing vaccination rates and reducing healthcare costs.
Are there any side effects?
The vaccine has a safety profile similar to existing mRNA vaccines, with common side effects including mild fever and soreness at the injection site.
Will it be available in other regions?
Following Europe's approval, other countries may consider authorizing the vaccine, depending on local regulatory processes and health needs.
How can healthcare providers prepare?
Providers should focus on staff training, proper storage, and patient education to ensure a smooth rollout of the new vaccine.
Key Takeaways
- Europe authorized the first mRNA combo vaccine for flu and COVID-19, a significant health milestone. Reuters reports on this milestone.
- Moderna's mRNA-1083 simplifies immunization by offering dual protection in one dose.
- Europe's proactive regulatory approval sets a precedent for global vaccination strategies.
- The U.S. regulatory process for combo vaccines is more cautious, delaying approval.
- Future trends include the expansion of combo vaccines to other viruses and innovative delivery methods.
- Healthcare providers must adapt to new vaccines with training and patient education.
- Public trust and clear communication are critical to overcoming vaccine hesitancy.
Related Articles
- AI-Driven Drug Discovery Enters Human Trials: A New Era in Medicine [2025]
- The Measles Outbreak in Idaho: Understanding the Risks and the Path Forward [2025]
- Understanding Peptides: A Comprehensive Guide [2025]
- Breaking Barriers: How Esther and Anne Wojcicki's New Healthcare Accelerator is Transforming the Industry [2025]
- Revolutionizing Memory: The Fusion of Synthetic DNA and Semiconductors [2025]
- The Misinterpretation of Diet Studies: Unraveling the Myths Linking Healthy Diets to Lung Cancer [2025]
![Europe Pioneers the First mRNA Combo Vaccine for Flu and COVID-19 [2025]](https://tryrunable.com/blog/europe-pioneers-the-first-mrna-combo-vaccine-for-flu-and-cov/image-1-1777066475594.jpg)



