Japan's Pioneering Move: The Future of Regenerative Medicine with Reprogrammed Cells [2025]
Introduction
Imagine a world where damaged tissues can be repaired, not just bandaged, where degenerative diseases are not a life sentence but a temporary setback. This vision is inching closer to reality with Japan's groundbreaking approval of the world's first treatments using reprogrammed human cells. Welcome to the future of regenerative medicine.
Japan's Ministry of Health, Labor, and Welfare recently granted conditional marketing authorization for two regenerative medical products derived from induced pluripotent stem cells (iPS cells). This approval, exactly 20 years after the creation of mouse i PS cells, signifies a monumental step in the practical application of i PS-derived products.
The global stem cell therapy market is expected to grow from
TL; DR
- Historic Approval: Japan is the first to approve iPS cell-derived treatments, heralding a new era in regenerative medicine.
- Potential Applications: iPS cells can revolutionize treatments for degenerative diseases, organ transplants, and more.
- Safety and Efficacy: Rigorous testing is crucial to ensure the safety and effectiveness of these treatments.
- Challenges Ahead: Ethical concerns, cost, and technical hurdles remain.
- Future Trends: Expect rapid advancements and broader applications in the next decade.
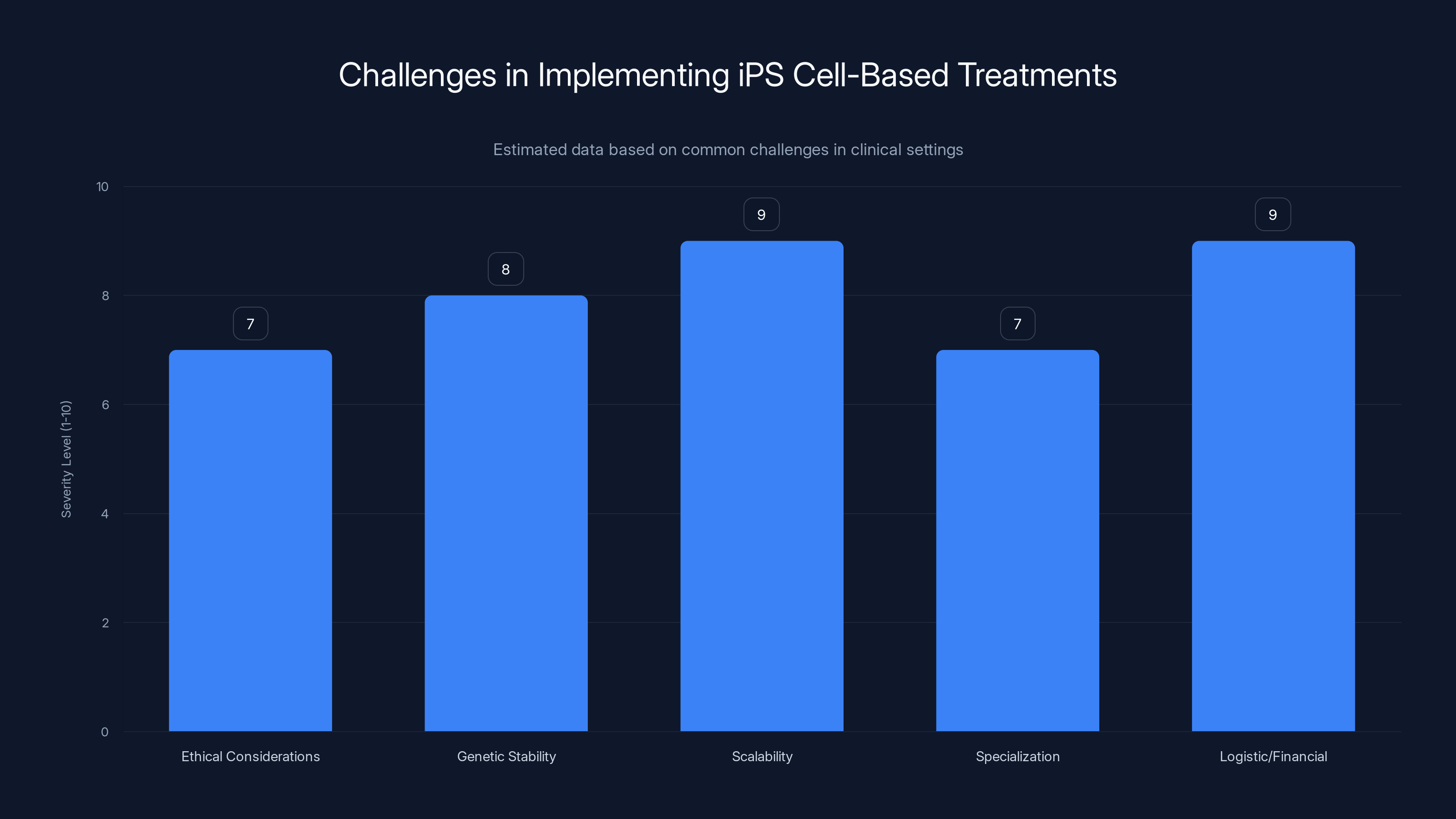
The most severe challenges in iPS cell-based treatments include scalability and financial logistics, with ethical considerations also being significant. Estimated data.
Unpacking Induced Pluripotent Stem Cells (i PS Cells)
Induced pluripotent stem cells, or iPS cells, are a type of stem cell that can be generated directly from adult cells. These cells are reprogrammed to an embryonic stem cell-like state by introducing factors that allow them to differentiate into any cell type in the body.
How i PS Cells Work
Creating iPS cells involves reprogramming somatic cells, like skin or blood cells, by introducing specific genes. This process effectively 'resets' the cells, giving them the ability to develop into various types of cells, such as neurons, heart cells, or pancreatic cells.
Applications of i PS Cells
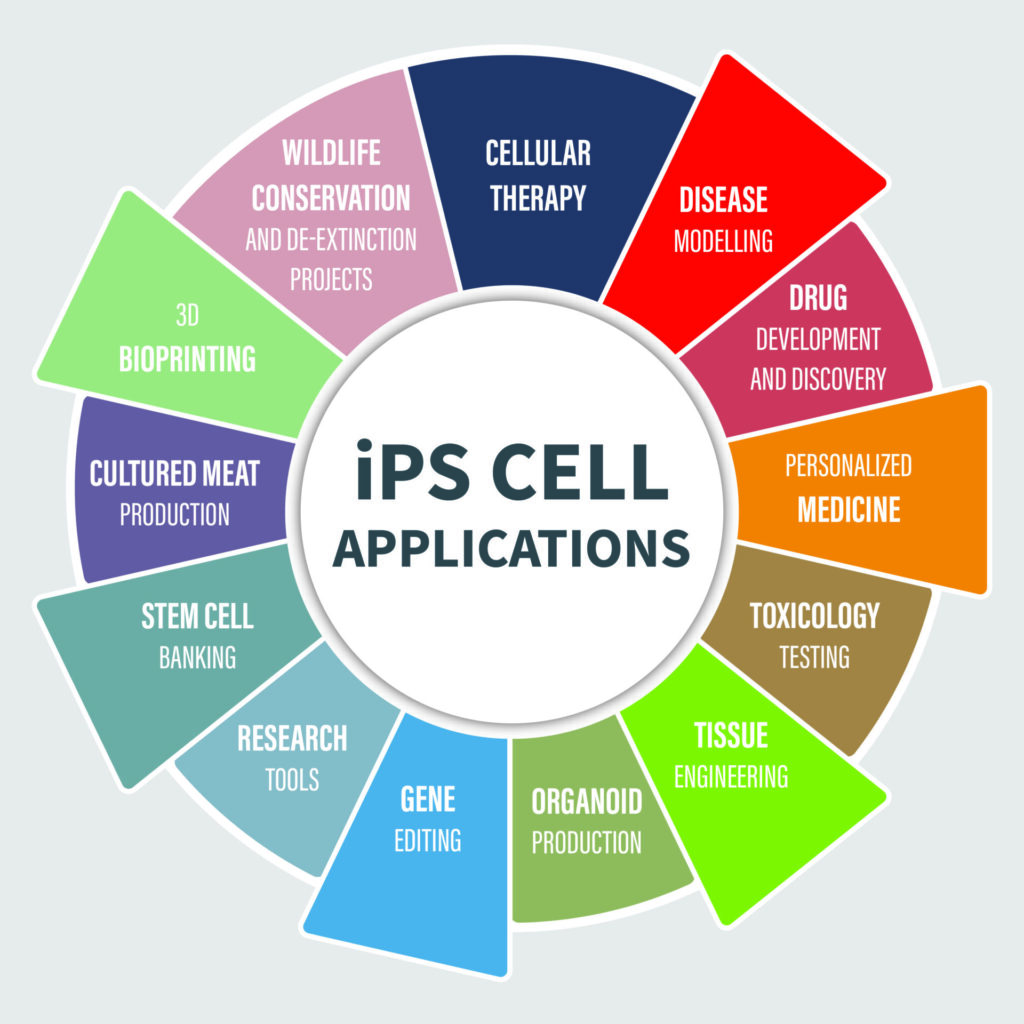
The potential applications of iPS cells are vast and varied, ranging from tissue repair and organ regeneration to disease modeling and drug testing. Here are a few key areas where iPS cells could make a significant impact:
- Regenerative Medicine: Repairing damaged tissues or organs, potentially eliminating the need for organ transplants.
- Disease Modeling: Creating disease-specific cells to study disease mechanisms and test new drugs.
- Personalized Medicine: Developing patient-specific cells that can be used for tailored treatments.
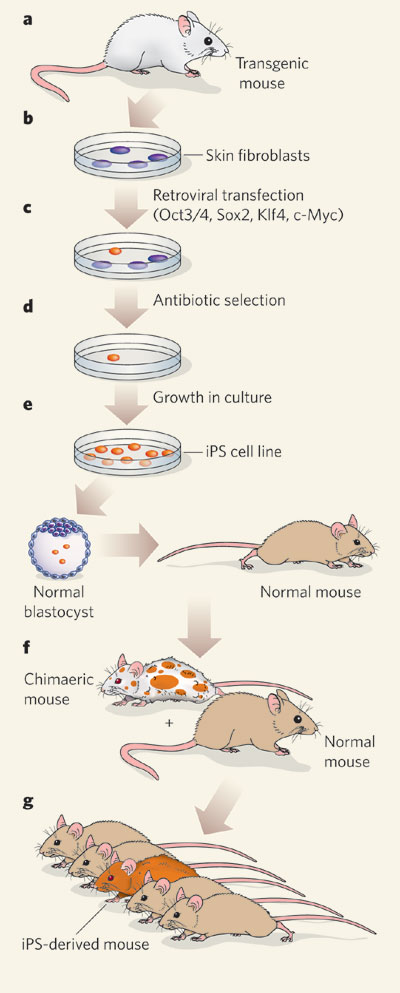
Japan's Approval: A New Era
The recent approval by Japan's Ministry of Health marks a significant milestone in the journey of iPS cells from the lab to clinical practice. This decision not only paves the way for new treatments but also sets a precedent for other countries to follow.
Why Japan?
Japan has long been at the forefront of iPS cell research, largely due to the pioneering work of Shinya Yamanaka, whose discovery of iPS cells earned him a Nobel Prize in 2012. Japan's regulatory environment, which supports rapid clinical application while ensuring safety and efficacy, has made it a global leader in this field.
The Approved Treatments
The two treatments approved involve the use of iPS cells to treat degenerative conditions. The first aims to restore vision in patients with retinal diseases, while the second targets spinal cord injuries.
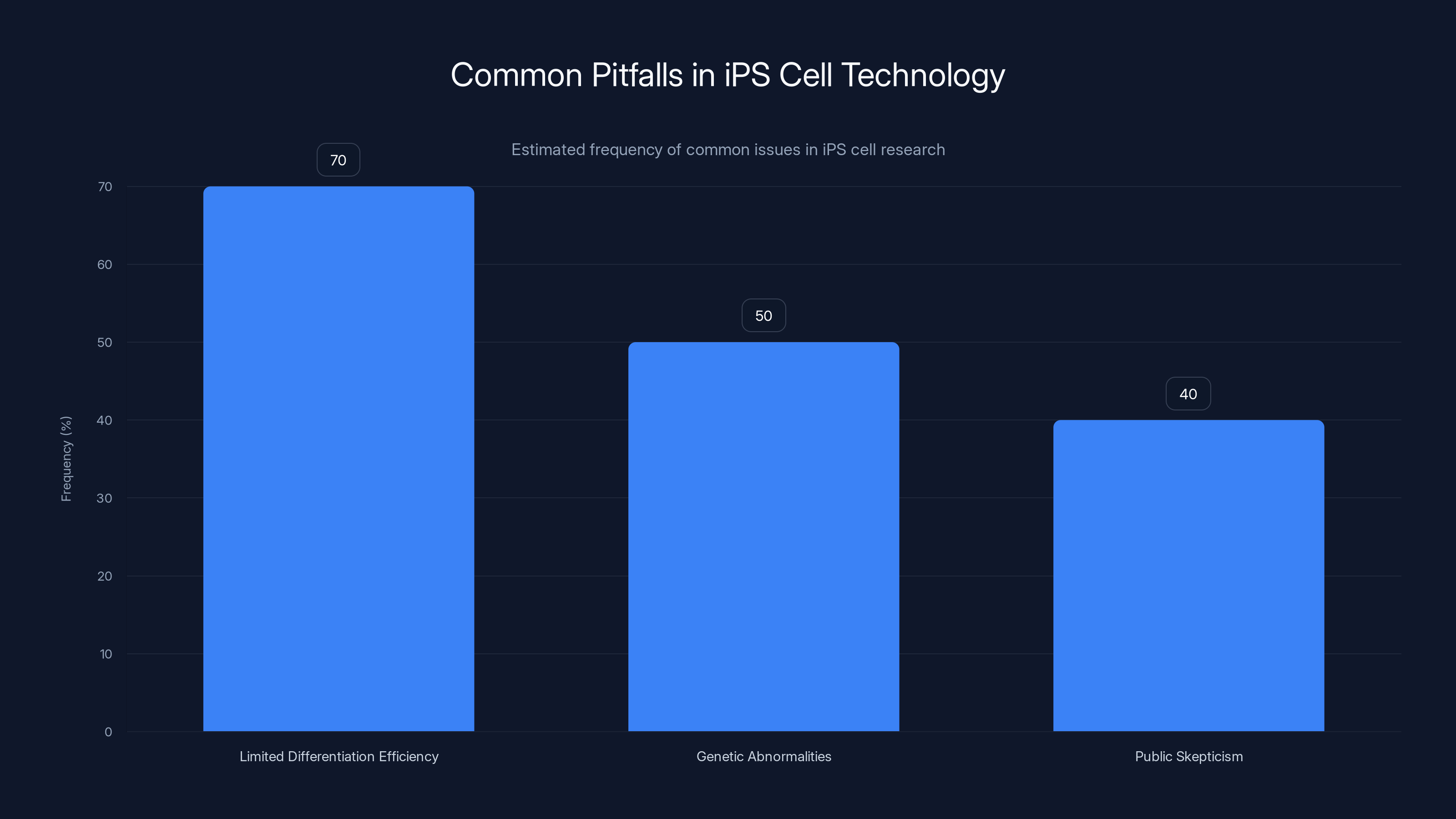
This chart estimates the frequency of common pitfalls in iPS cell technology, highlighting that limited differentiation efficiency is the most prevalent issue. Estimated data.
Practical Implementation and Challenges
Implementing iPS cell-based treatments in clinical settings poses several challenges, from ethical considerations to technical and logistical hurdles.
Ethical Considerations
The use of stem cells, particularly those derived from human sources, raises ethical questions. Ensuring informed consent from donors and addressing concerns about genetic manipulation are crucial.
Technical Challenges
Producing iPS cells that are safe and effective requires overcoming several technical barriers:
- Genetic Stability: Ensuring that reprogrammed cells maintain genetic integrity over time.
- Scalability: Developing methods to produce iPS cells at a scale suitable for clinical use.
- Specialization: Directing iPS cells to develop into the desired cell types efficiently.
Logistic and Financial Considerations
The cost of iPS cell production and the infrastructure required for their clinical application present significant financial challenges. Governments, private investors, and research institutions must collaborate to fund research and development.
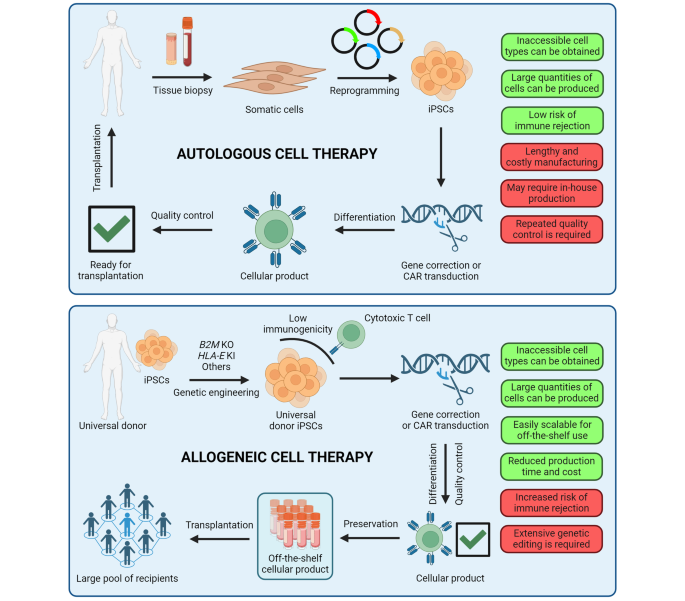
Future Trends in Regenerative Medicine
As iPS cell technology matures, several trends are likely to shape its future.
Broadening Applications
Expect to see iPS cell technology applied to a wider range of diseases, including neurodegenerative disorders, diabetes, and cardiovascular diseases.
Integration with Other Technologies
The integration of iPS cells with technologies like CRISPR gene editing and artificial intelligence will accelerate the development of new treatments.
Global Regulatory Harmonization
As more countries approve iPS cell treatments, a global regulatory framework may emerge, facilitating international collaboration and standardization.

Best Practices for Researchers and Clinicians
For those working in the field, adhering to best practices is crucial to advancing iPS cell research and its clinical application.
Establishing Robust Protocols
Developing standardized protocols for iPS cell production and differentiation is essential to ensure reproducibility and safety.
- Quality Control: Implement rigorous quality control measures at every stage of iPS cell production.
- Documentation: Maintain detailed records of cell lines, including their genetic profiles and differentiation pathways.
Collaboration and Data Sharing
Fostering collaboration among researchers and sharing data openly can accelerate progress and avoid duplication of efforts.
Common Pitfalls and Solutions
Navigating the complexities of iPS cell technology can be challenging. Here are some common pitfalls and strategies to avoid them:
Overcoming Technical Hurdles
-
Problem: Limited differentiation efficiency.
-
Solution: Optimize culture conditions and use small molecules to enhance differentiation.
-
Problem: Genetic abnormalities in iPS cells.
-
Solution: Regularly screen cell lines for genetic stability and employ non-integrating vectors for reprogramming.
Addressing Ethical Concerns
- Problem: Public skepticism about genetic manipulation.
- Solution: Engage in public education and transparent communication about the benefits and risks of iPS cell technology.
Conclusion
Japan's approval of iPS cell-derived treatments marks a significant leap forward in regenerative medicine. As this technology continues to evolve, it promises to transform the landscape of healthcare, offering new hope for patients with previously untreatable conditions.
The journey from discovery to clinical application is fraught with challenges, but the potential rewards are immense. By embracing innovation, collaboration, and ethical responsibility, the scientific community can unlock the full potential of iPS cells, paving the way for a healthier future for all.
FAQ
What are iPS cells?
Induced pluripotent stem cells (iPS cells) are adult cells that have been genetically reprogrammed to an embryonic stem cell-like state, allowing them to develop into any cell type in the body.
How do iPS cells differ from embryonic stem cells?
Unlike embryonic stem cells, iPS cells are derived from adult tissues, which avoids ethical issues related to the use of embryos.
What diseases can be treated with iPS cells?
iPS cells have the potential to treat a wide range of diseases, including degenerative disorders, cardiovascular diseases, and diabetes.
What are the risks associated with iPS cell treatments?
Potential risks include genetic instability and the possibility of tumor formation. Rigorous testing and quality control are essential to mitigate these risks.
How are iPS cells produced?
iPS cells are created by introducing specific genes into adult cells, reprogramming them to a pluripotent state.
What is the future of iPS cell technology?
The future of iPS cell technology lies in its integration with other technologies like gene editing and AI, expanding its applications and improving treatment outcomes.
Key Takeaways
- Japan's approval of iPS cell-derived treatments is a world first, marking a pivotal moment in regenerative medicine.
- iPS cells offer transformative potential for treating degenerative diseases and personalizing medicine.
- Ensuring the safety and efficacy of iPS cell treatments requires rigorous testing and quality control.
- Ethical, technical, and financial challenges must be addressed to advance iPS cell technology.
- The integration of iPS cells with gene editing and AI will drive future advancements in healthcare.
Related Articles
- Unmasking Sleep Apnea in Women: Understanding, Detecting, and Treating the Silent Epidemic [2025]
- How Technology Is Revolutionizing Sleep Apnea Treatment [2025]
- Amazon Expands Healthcare AI Assistant Across Its Platform [2025]
- Revolutionizing Women's Health: Whoop's New Blood Test Initiative [2025]
- Measles Resurgence: A Grim Warning for Future Pandemics [2025]
- AI Science Startups: Securing the Future with $114M in Funding [2025]
![Japan's Pioneering Move: The Future of Regenerative Medicine with Reprogrammed Cells [2025]](https://tryrunable.com/blog/japan-s-pioneering-move-the-future-of-regenerative-medicine-/image-1-1773480841454.jpg)



