Restoring the American Chestnut: Modern Genetics Brings Back a Forest Icon
When your great-grandfather walked through an Appalachian forest in 1900, nearly one in every four trees was an American chestnut. These weren't just any trees. They were giants, reaching 100 feet tall, with trunks so thick it took three people holding hands to wrap around them. They dominated the canopy, defined the ecosystem, and shaped how humans lived in those mountains. Then, within just 50 years, they were gone.
We're not talking about a slow decline. An estimated 3 billion American chestnuts died, wiped out by two invasive fungal pathogens that arrived from Asia in the early 1900s. For most of the 20th century, the species existed only in botanical memory and scattered stumps that kept trying—and failing—to regrow. But something changed in the last two decades. Scientists started asking a question that would have seemed impossible 30 years ago: What if we could bring them back?
The answer turns out to be far more complicated than any single approach. It's not just about finding disease-resistant trees or breeding them together. It's about combining cutting-edge genomics, centuries-old agricultural techniques, genetic modification, and raw determination. In February 2026, researchers from the American Chestnut Foundation published their most comprehensive findings yet in Science, describing a decade-long experiment involving over 4,000 trees, millions of data points, and three fundamentally different strategies to restore an ecosystem that's been broken for longer than most of us have been alive.
This is the story of how science is trying to resurrect a ghost from the forest.
TL; DR
- 3 billion American chestnuts died from two Asian fungi that arrived in the early 1900s, making the species "functionally extinct"
- Three restoration strategies are being tested: breeding naturally resistant American chestnuts, creating hybrid trees with disease-resistant Chinese chestnuts, and genetic engineering to enhance fungal resistance
- Researchers tested over 4,000 trees for over 14 years, finding that genetically modified trees face unexpected growth penalties but some show promise
- Natural resistance does exist in surviving American chestnuts, but it's rarer and weaker than hoped, requiring careful breeding programs
- Timeline is measured in decades, not years, making this one of the longest biological restoration projects ever attempted

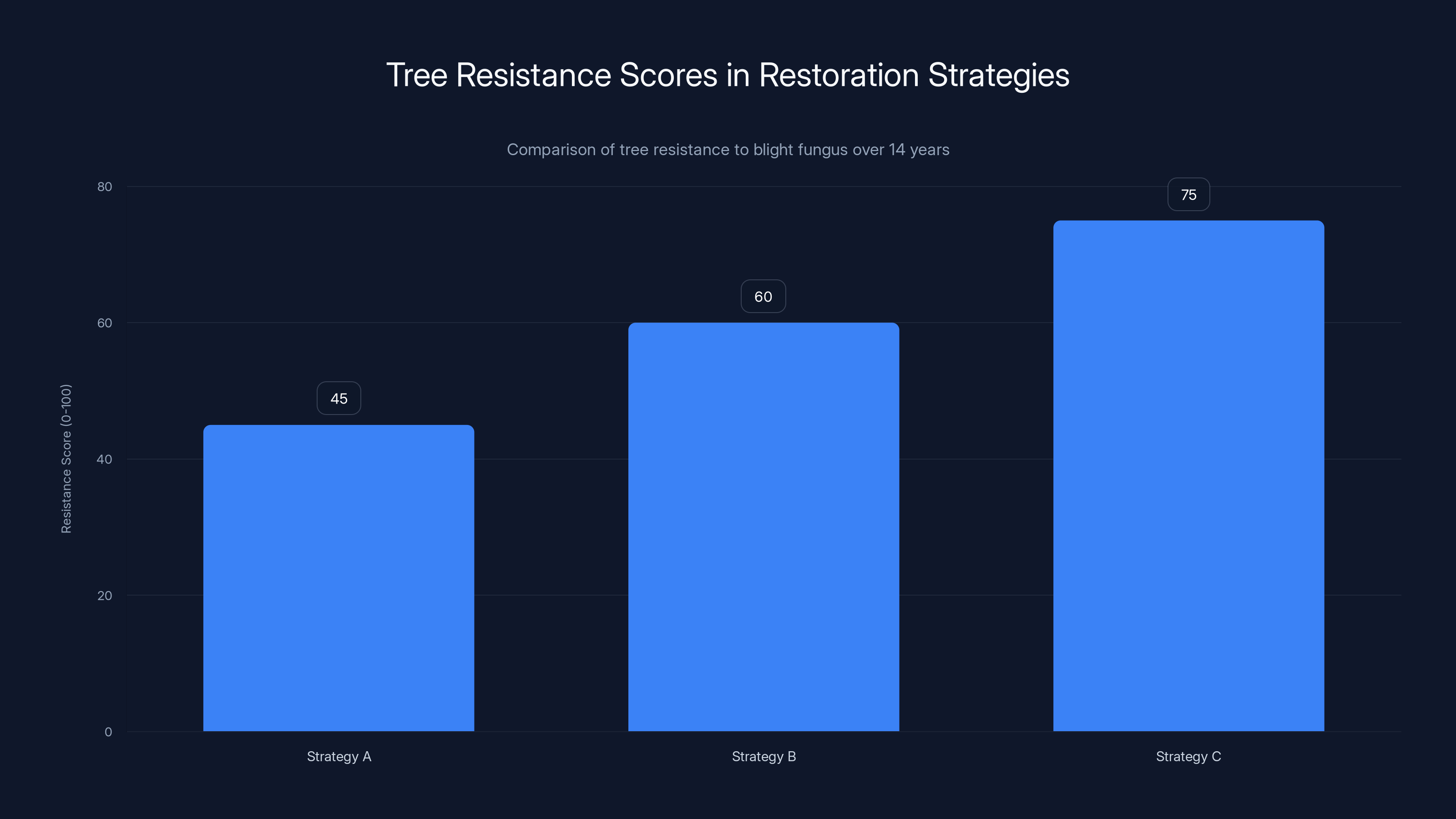
Estimated data shows Strategy C had the highest average resistance score, indicating better adaptation to the blight fungus over 14 years.
The Catastrophe That Nearly Erased a Species
To understand what happened to the American chestnut, you need to picture the eastern forests as they existed in 1890. Not as a diverse ecosystem with many dominant species, but as something closer to a monoculture—one that nature created rather than agriculture. Chestnuts made up between 25 and 40 percent of trees in hardwood forests from New England to the Carolinas. In some valleys of Appalachia, the percentage was even higher.
These weren't marginal trees that could be replaced. American chestnuts were the bread and butter of the forest ecosystem. They produced nuts that fed wildlife from passenger pigeons (now extinct themselves) to bears, deer, and squirrels. Their wood was straight-grained and rot-resistant, making them ideal for construction and railroad ties. American families harvested chestnuts in the fall like modern people visit pumpkin patches—it was a cultural touchstone.
Then came 1904. A forester noticed dying chestnut trees in the Bronx Zoo. Within a few years, the problem was unmistakable: an aggressive fungus called Cryphonectria parasitica, commonly known as chestnut blight, was spreading through the forests at an alarming rate. The fungus, which had co-evolved with Asian chestnut species and rarely killed them, found American chestnuts had virtually no defense. The blight would infect the tree's bark and girdle it, cutting off nutrient flow until the tree died.
By 1950, the American chestnut was functionally extinct in its native range—meaning you could search an entire forest and find no reproducing population, only sprouts growing from old root systems that died before reaching maturity. The ecosystem that had been shaped by millions of years of evolution was suddenly transformed. Oak trees moved in to fill the gap. The energy flow through the food web shifted. Species that depended on chestnut nuts declined or disappeared.
If the story had ended there, it would be just another example of ecological collapse from invasive species. But it didn't end there.
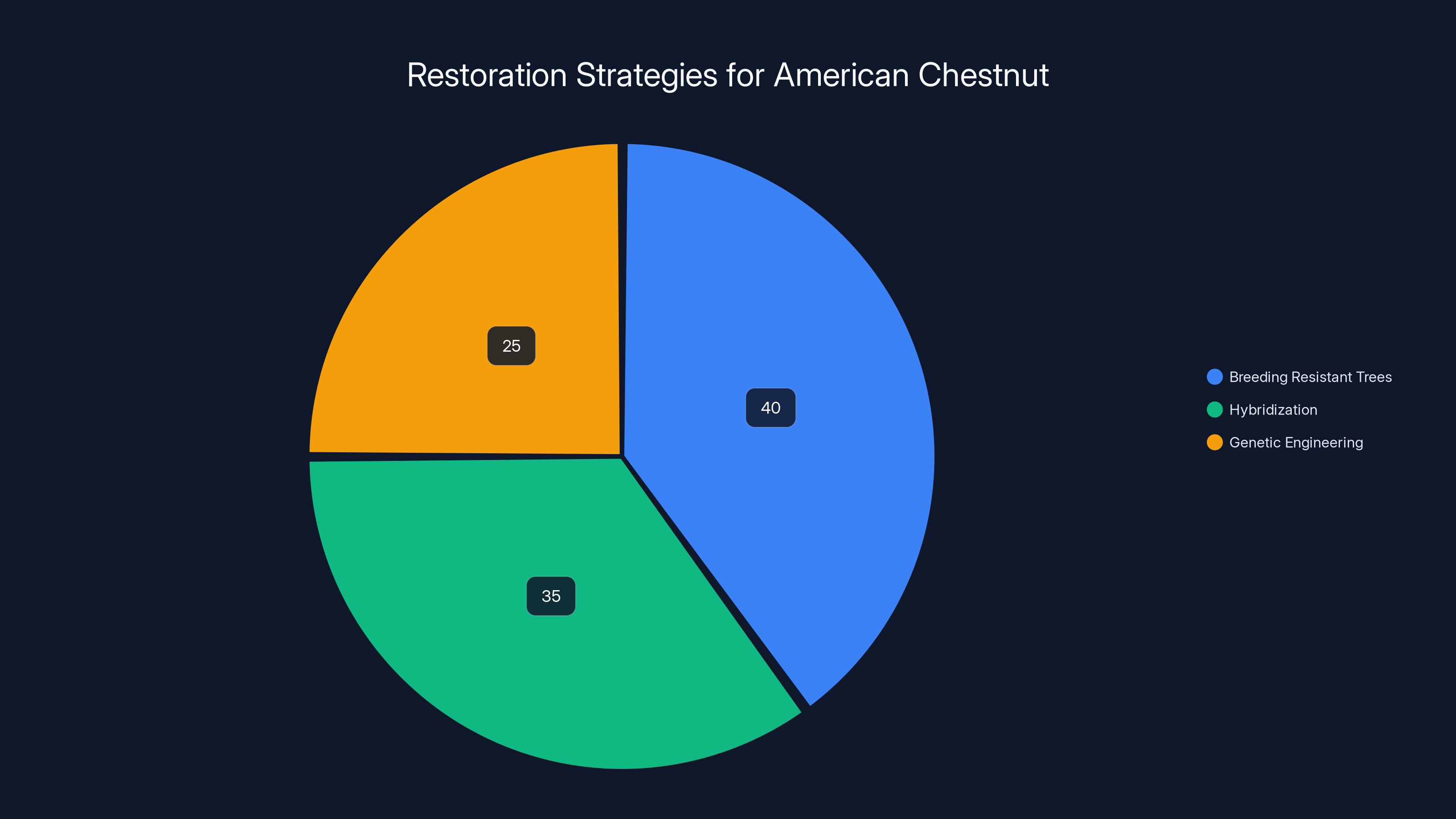
Efforts to restore the American chestnut are distributed among breeding resistant trees (40%), hybridization (35%), and genetic engineering (25%). Estimated data.
Why Complete Extinction Didn't Happen
The American chestnut survived in fragments. Not in Appalachia, but in drier regions of western North America where the fungal pathogens couldn't thrive. Botanical gardens, private collections, and some planted groves in the Southwest maintained living populations. These weren't thriving populations, but they were populations nonetheless.
More importantly, a few individual trees seemed to possess some inherent resistance to the blight. These anomalies didn't make sense at first. The tree would be growing in conditions identical to other chestnuts nearby, yet it would survive an infection that killed everything around it. Scientists started referring to these as Large Surviving American Chestnuts, or LSACs. They became the cornerstone of one restoration strategy.
A second survival pathway came from an unexpected source: a virus that attacked the blight fungus itself. In certain regions where this virus was present, chestnut trees could actually coexist with the blight without dying. The virus would weaken or kill the fungus, keeping infection rates low enough for the tree to survive. This suggested that biological control—introducing a disease of the disease—might be part of the solution.
Finally, there was the Chinese chestnut. While American chestnuts had evolved completely defenseless against the Asian fungus, Chinese chestnuts had evolved alongside it. They carried genetic resistance that had been refined over thousands of years of co-evolution. Chinese chestnuts weren't great trees for the American landscape—they were smaller, their wood was less valuable, their form didn't match the ecological role American chestnuts had filled. But they could survive.
What if you could move the resistance from the Chinese chestnut into the American chestnut's genome? That question led researchers down three parallel paths.

Strategy One: Breeding Naturally Resistant American Chestnuts
The first strategy seems deceptively simple: find the American chestnuts that show natural resistance and breed them together. If you could identify even a few resistant individuals and crossbreed them strategically, you might create offspring that inherited these resistance traits. Over multiple generations, you could potentially concentrate resistance while maintaining the American chestnut's other characteristics—its growth rate, wood quality, and ecological fit.
The problem is that trees aren't like mice. You can't run generational breeding experiments quickly. An American chestnut takes 5-10 years before it produces viable seeds. If you want to evaluate the progeny's resistance to the fungus, you need to infect them and then wait years for the infection to develop and the tree to either survive or die. A single breeding cycle that would take a few months in a fruit fly takes a decade in a chestnut.
But the real problem was uglier than that: there wasn't much natural resistance to work with. When researchers systematically tested the surviving American chestnuts they could find, most showed only modest resistance. On a 0-to-100 scale where higher numbers meant better survival after infection, most LSACs scored in the 30-50 range. Out of 143 surviving American chestnut trees that researchers tested, only seven had resistance scores above 50.
This meant that even the most resistant American chestnuts were still susceptible. They might survive longer than a normal tree, but they probably wouldn't thrive in an environment where the fungus was actively present. To create a tree that could truly return to Appalachia, you'd need to find a way to boost that resistance further. Interbreeding the most resistant trees might help, but the math was discouraging. You'd be combining imperfect traits across a long timeline.
Yet researchers pursued this strategy anyway. Because breeding resistant trees had one crucial advantage: any trees produced would be 100 percent American chestnut genetically. There would be no hybrid genetics, no transgenic modifications to debate. Just American chestnut, evolved through classical selective breeding.
This approach remains ongoing. Scientists continue to identify resistant American chestnuts, establish breeding populations, and track their offspring. Progress is slow but steady. The hope is that over a sufficiently long timeline—possibly 20-30 more years—breeding could generate trees with high resistance.
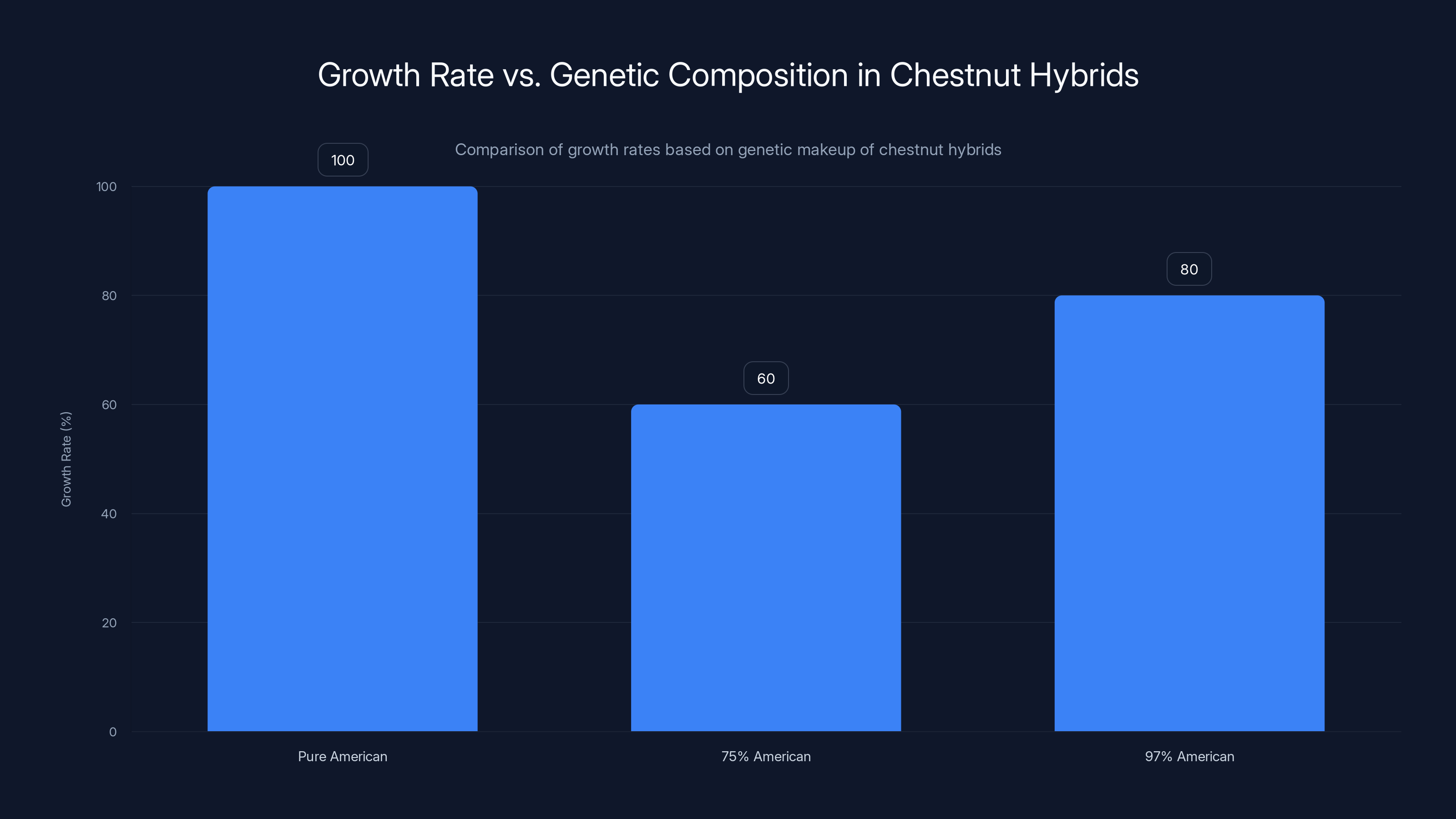
Estimated data: Pure American chestnuts grow at full rate, while hybrids with 75% and 97% American genetics grow at 60% and 80% of that rate, respectively.
Strategy Two: Creating Hybrid Vigor Through Backcrossing
The second strategy took a different approach: start with a hybrid. Cross an American chestnut with a Chinese chestnut to create a F1 hybrid. The hybrid would inherit the Chinese chestnut's fungal resistance and the American chestnut's general form. Then, breed that hybrid back to a pure American chestnut. Then breed that offspring back to an American chestnut again. And again. And again.
This process, called backcrossing, sounds straightforward in theory. Each generation you're removing Chinese chestnut genes and adding back American chestnut genes. After enough backcrosses—typically 5-10 generations—you should end up with a tree that's 97-99 percent American chestnut genetically but carries the crucial Chinese chestnut resistance genes.
The advantage over pure breeding programs is speed. You're not waiting for resistance to emerge from within the American chestnut population. You're directly incorporating proven resistance from a different species. The disadvantage is that you're introducing non-native genetic material, which raises ecological and philosophical questions. Some people argue that a 97 percent American chestnut with 3 percent Chinese genetics isn't really American chestnut restoration—it's creating a new hybrid species.
When researchers tested backcrossed hybrids, they found something surprising: these trees often grew more slowly than pure American chestnuts in healthy conditions. A hybrid that was 75 percent American chestnut and 25 percent Chinese chestnut might have excellent fungal resistance but only grow at 60 percent the rate of a pure American chestnut. This matters because the American chestnut's historical dominance in the canopy was partly due to its rapid growth. A slow-growing chestnut wouldn't regain ecological dominance the way the original species had.
Researchers were stuck with a trade-off: they could have resistance or growth rate, but combining both traits seemed difficult. The genetic basis for this trade-off wasn't fully understood, but it suggested that resistance and rapid growth might be controlled by competing genetic pathways.
Despite these challenges, backcrossing programs continue. The University of Massachusetts, the State University of New York College of Environmental Science and Forestry, and the American Chestnut Foundation maintain large populations of hybrid chestnuts at various backcross generations. The theory is sound—eventually, selection and recombination should produce trees with both traits—but the timescale is measured in decades.
Strategy Three: Genetic Engineering and Transgenic Chestnuts
The third strategy bypasses breeding altogether. Scientists identified oxalic acid as one of the key weapons the blight fungus uses to attack chestnut trees. The fungus secretes oxalic acid, which damages the tree's cellular tissues and allows the infection to spread. Meanwhile, wheat plants naturally produce an enzyme that breaks down oxalic acid, neutralizing this fungal weapon.
Researchers took the wheat gene that codes for this enzyme and inserted it directly into the American chestnut genome, creating a genetically modified tree that could potentially disarm one of the blight's main attack mechanisms. If the approach worked, you'd have a tree that was 100 percent American chestnut, with no hybrid genetics, just an added tool to fight the fungus.
The appeal was obvious. In theory, you could create blight-resistant American chestnuts without the multi-decade breeding timeline. You wouldn't need to compromise with other species' genetics or accept slower growth. Just enhance the American chestnut's natural defenses and let it return to the forest.
The reality was messier. When researchers tested genetically modified chestnuts, they discovered a problem with the gene insertion itself. The wheat gene had inserted into an important region of the chestnut genome—specifically into a gene that's crucial for plant growth in many species. Trees that inherited two copies of the inserted gene survived at only 16 percent of their expected rate. Even trees with a single copy grew about 22 percent slower than unmodified chestnuts.
This wasn't a theoretical problem. It was a real biological constraint. The very trait you wanted—oxalic acid degradation—was linked to reduced growth and survival in these trees. The genetic engineering approach had created trees that could fight the fungus but couldn't grow properly.
But—and this was important—the variability was huge. Most genetically modified trees had severe growth penalties. But a small percentage, around 4 percent of the tested trees, showed both high blight resistance and growth rates comparable to unmodified American chestnuts. These exceptional individuals proved that the combination was theoretically possible. The challenge was figuring out why some trees handled the transgenic modification so much better than others.
Researchers hypothesized that genetic background mattered. The position where the gene inserted might vary slightly in different American chestnut lines, or other genes elsewhere in the genome might modify how much the growth penalty appeared. They began planning second-generation experiments where they could select only from American chestnut lines that tolerated the wheat gene insertion better.

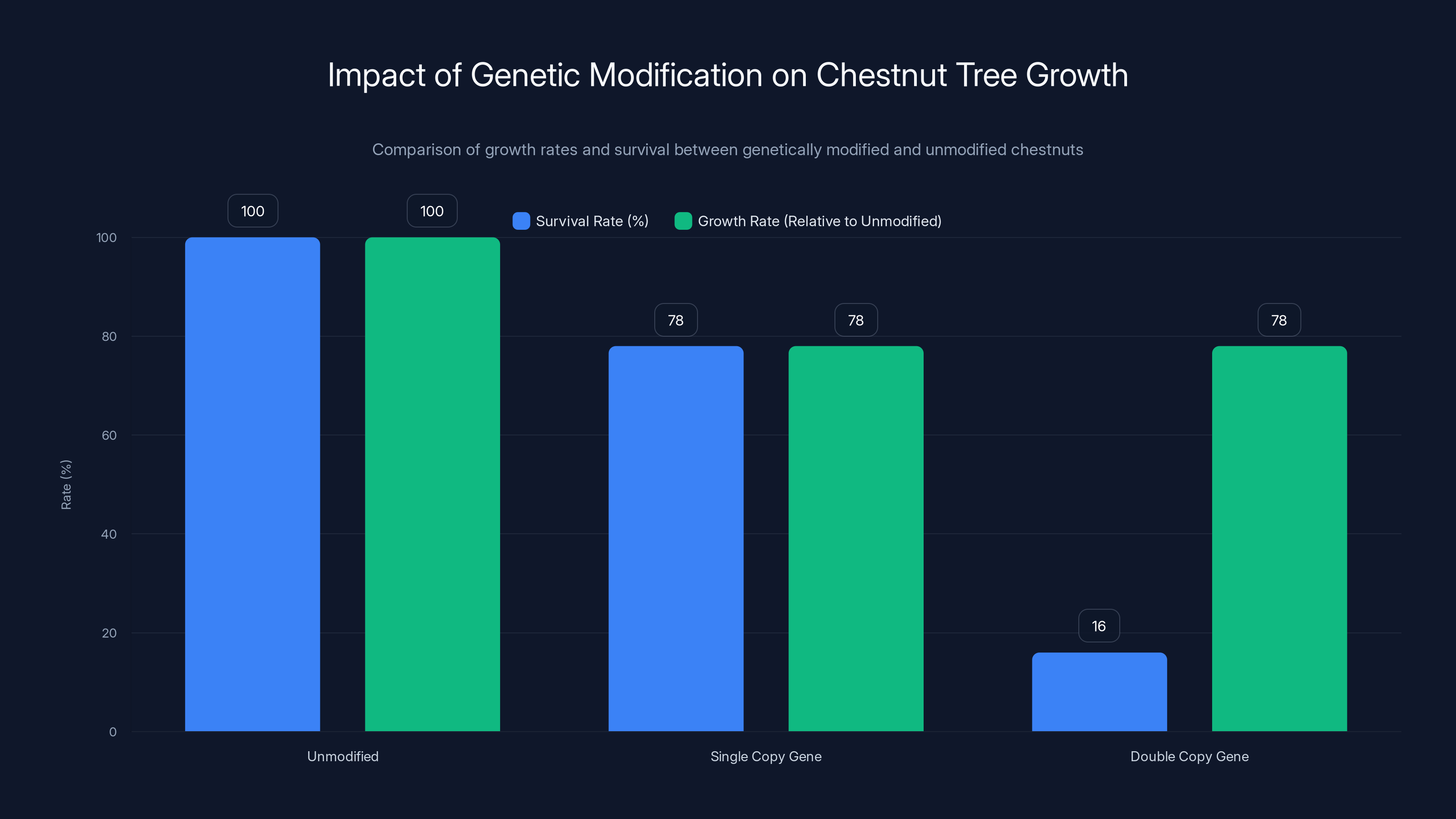
Genetically modified chestnuts with a single copy of the wheat gene grew 22% slower, while those with two copies had a survival rate of only 16%. Estimated data based on narrative.
The American Chestnut Foundation's Comprehensive Testing Framework
In 2012, researchers affiliated with the American Chestnut Foundation set out to do something unprecedented: systematically compare all three restoration strategies side-by-side using rigorous, quantifiable methods. They weren't trying to prove one approach was superior. They were trying to understand the actual biology and genetics underlying each approach so that decisions could be made on evidence rather than hope.
The scale of the effort was staggering. Over 14 years, researchers infected more than 4,000 individual trees with the blight fungus under controlled conditions. They planted these trees in Appalachian nurseries to simulate real-world growing conditions. They tracked survival, growth rate, infection severity, and recovery over years of observation. The trees were scored on a 0-to-100 scale based on how much tissue damage the fungal infection caused. A score of 0 meant no damage, essentially complete resistance. A score of 100 meant the tree was killed by the infection.
Parallel to the field trials, researchers conducted genomic sequencing, generating the highest-quality genomes of both American and Chinese chestnuts ever assembled. They performed biochemical assays to understand how trees responded to infection at the molecular level. They measured gene expression patterns to see which genes activated in response to the fungus. They tested for the presence of the blight-suppressing virus in different tree populations.
The data generated was massive—millions of individual measurements from thousands of trees. This wasn't casual observation. This was hypothesis-driven science with controls, replicates, and statistical validation.
One key finding emerged immediately: survival in the face of the fungus required active adaptation. Trees couldn't just be passively resistant. When infection began, their cells needed to mount a response—producing compounds that fought the fungus, compartmentalizing infection, repairing damaged tissue. Trees that mounted a stronger response survived longer. But this response came at a cost. Fighting an infection is metabolically expensive. A tree mounting a strong immune response was a tree burning energy that could have gone into growth.
Another finding was about timing. In controlled greenhouse conditions where researchers inoculated trees with the fungus, results varied significantly based on temperature and humidity. The blight fungus grows more aggressively in warm, humid conditions. At cooler temperatures, the same fungal strain developed more slowly, giving trees more time to mount a response. This suggested that in Appalachia's cooler mountain regions, the same genetic resistance might be more effective than in warmer lowland areas.

The Mystery of Slow-Growing Resistance
One of the most puzzling findings came from the hybrid backcross lines. When researchers planted Chinese and Chinese-American hybrid chestnuts in areas where a virus suppressed the blight fungus, allowing trees to grow without constant fungal pressure, the hybrids consistently grew slower than pure American chestnuts. Chinese chestnuts are smaller-statured trees naturally, and the hybrids seemed to inherit that characteristic.
But why? The researchers had anticipated that in a disease-free environment, genetic resistance shouldn't carry a growth penalty. A tree with blight resistance wouldn't need to mount a constant immune response if the blight wasn't present. It should grow normally.
The answer seemed to be genetic incompatibility at a fundamental level. American and Chinese chestnuts had diverged evolutionarily for millions of years. Their developmental programs—the genetic instructions that governed how they grew from seedling to mature tree—weren't perfectly compatible. A hybrid inheriting genes from both species sometimes had conflicting instructions. One set of genes might be pushing for rapid growth while another set was tuning growth more slowly. The result was a compromise growth rate.
Some hybrid lines showed this effect more severely than others, suggesting that specific genetic combinations mattered. A hybrid that inherited certain American chestnut genes along with the Chinese resistance genes might grow better than one that inherited different American genes. This meant that future backcrossing programs could potentially select for hybrids that combined good growth with good resistance—but it would require careful tracking of which American chestnut genetics were being carried through each generation.

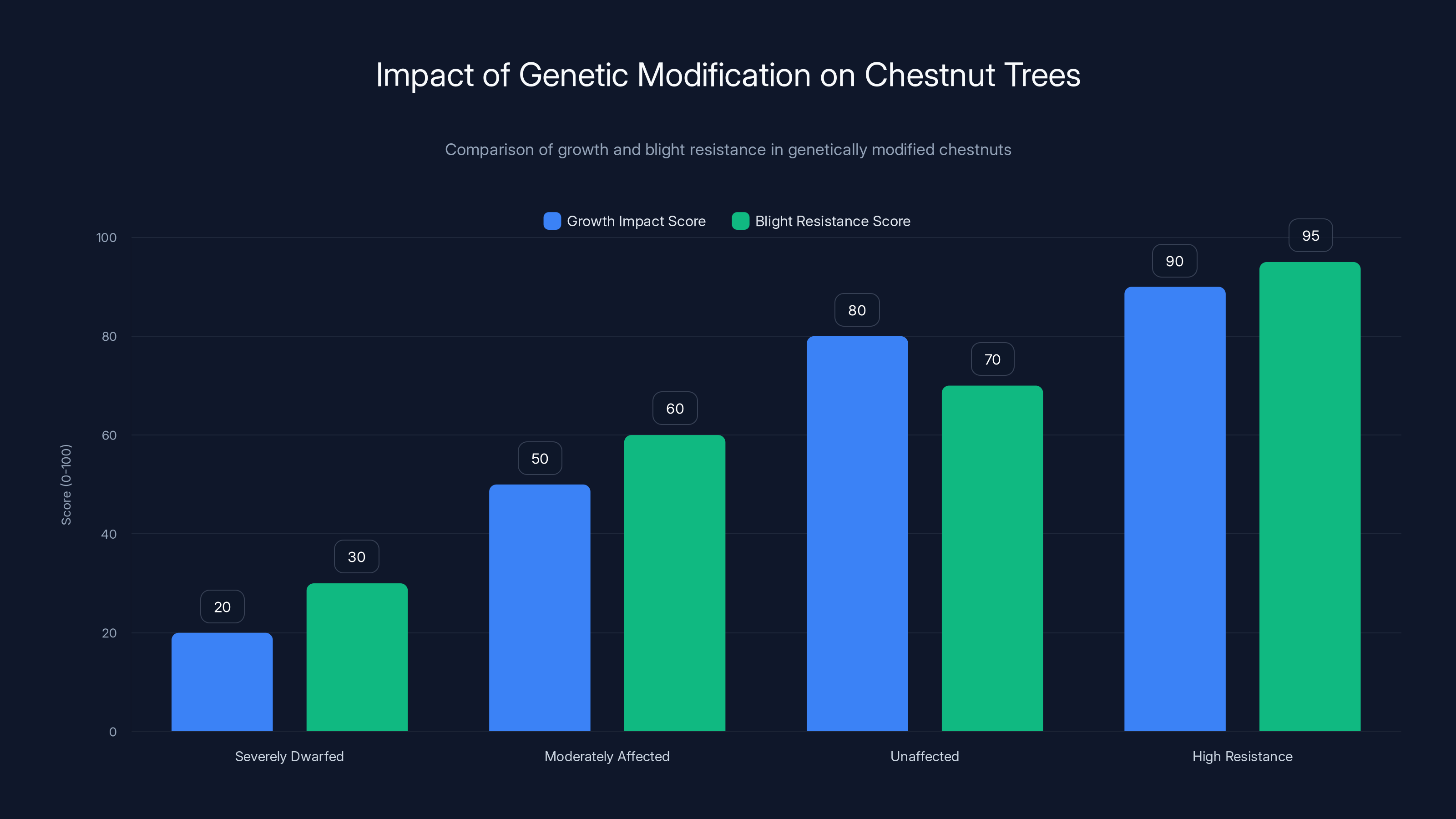
Estimated data shows variation in growth and blight resistance among genetically modified chestnuts. Only 4% of trees achieved high resistance with normal growth.
Genetic Engineering's Unforeseen Consequences
The genetically modified American chestnuts revealed another lesson about the complexity of genetic systems. Researchers had inserted the oxalic-acid-degrading wheat gene into the chestnut genome, assuming it would simply add a new function to the chestnut's existing capabilities. Fight the fungus's main weapon, and the chestnut should survive better.
What they hadn't fully anticipated was how sensitive plant growth is to disruption. The gene happened to insert near or within a gene that's crucial for normal plant development. In many plant species, this particular gene family controls how cells divide and how tissues differentiate. Disrupt it, and you get developmental problems—stunted growth, reduced vigor, altered root systems.
The severity of the penalty varied enormously among individual transgenic trees. Some were severely dwarfed. Others barely showed any growth difference from wild-type chestnuts. This variation suggested that either the insertion site differed among trees, or genetic background differences modified how much the disruption mattered.
Researchers identified one small population of transgenic trees—about 4 percent of those tested—that showed an exceptional combination of traits. These trees had high resistance to blight, as evidenced by a high score on the 0-to-100 damage scale, yet they maintained growth rates within normal range for American chestnuts. Why these specific trees managed this balance was unclear. It might have been luck in where the gene inserted. It might have been that their specific genetic background somehow buffered the negative effects. It might have been compensatory mutations elsewhere in the genome.
The implication was important: genetic engineering wasn't a dead-end approach. It was just more complicated than initially hoped. Future attempts might succeed if researchers could better control where the gene inserted in the genome, or if they could breed for genetic backgrounds that tolerated the insertion better, or if they could engineer the gene itself to have less collateral damage.

Understanding Growth Rate as an Evolutionary Constraint
One theme emerged across all three restoration strategies: growth rate kept coming up as a limiting factor. Natural selection had shaped American chestnuts to grow quickly—this was probably essential for their dominance in the canopy. Any restoration effort that compromised growth rate would struggle.
With the pure breeding approach, the problem was straightforward: the most resistant American chestnuts, by and large, didn't grow faster. The genes for rapid growth and fungal resistance seemed to be independent in American chestnut populations. You couldn't find an American chestnut that had both traits unless they appeared together by chance.
With hybrids, the problem was genetic incompatibility. Chinese and American chestnut developmental genetics didn't perfectly align. A hybrid tree had to navigate between two different sets of developmental instructions.
With genetic engineering, the problem was that adding a new function disrupted an existing system. The wheat gene insertion happened to hit a growth-controlling gene.
Researchers began to suspect that the relationship between resistance and growth might be fundamentally constrained. Perhaps there was a biological trade-off at the physiological level. An organism investing heavily in maintaining an active immune system might have less energy available for growth. Or perhaps the genetic pathways that confer fungal resistance are naturally slow-growth pathways, linked by developmental history.
This suggested that restoration chestnuts—regardless of which strategy succeeded—might never grow quite as fast as the original pre-blight American chestnuts had. They might be good enough to compete in the forest canopy, but they might not achieve the same dramatic dominance. The ecosystem they returned to would be different from the ecosystem they had left.

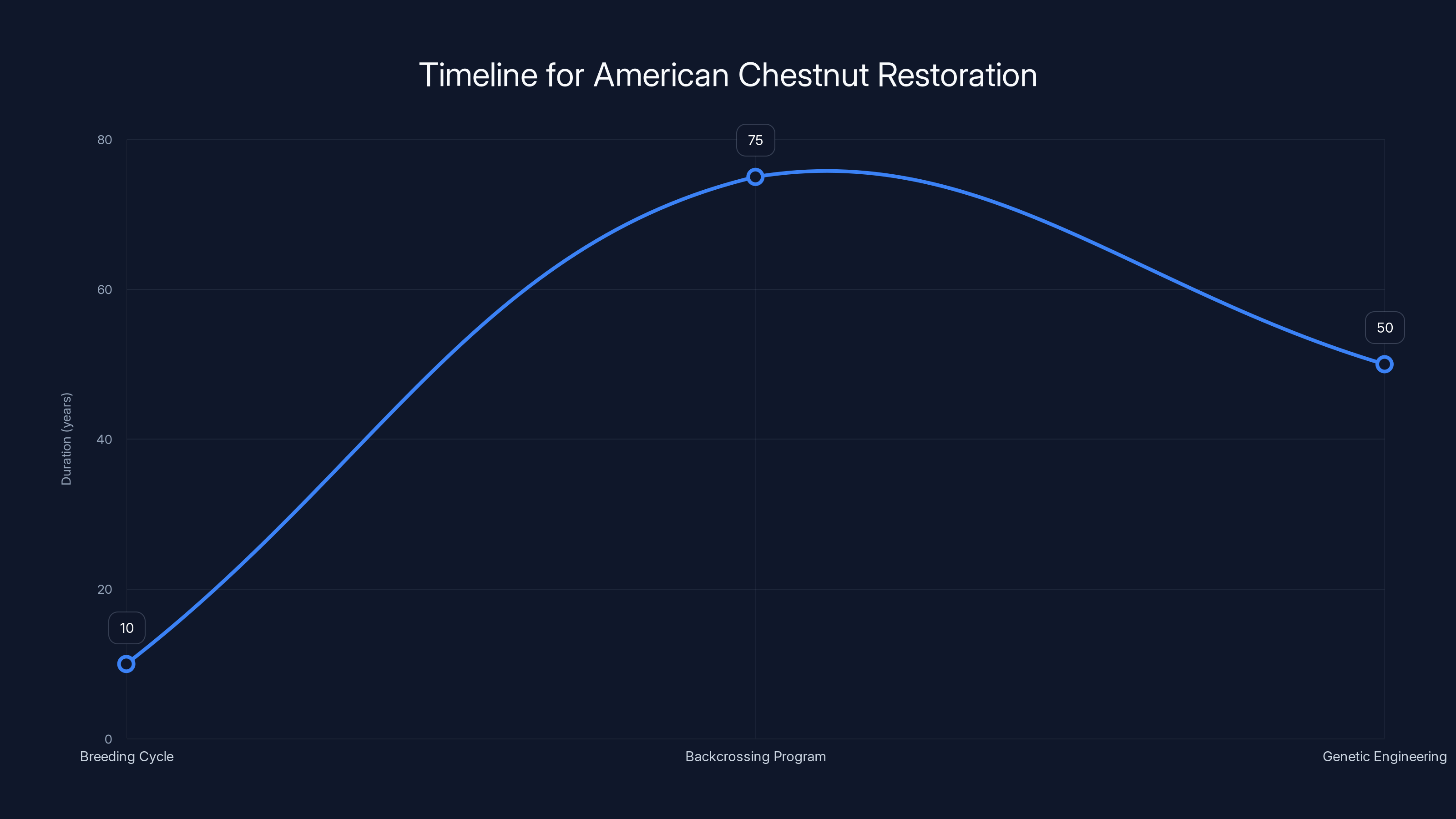
The restoration of American chestnuts through traditional breeding and genetic engineering spans decades, with breeding cycles taking 10 years, backcrossing programs 75 years, and genetic engineering 50 years. Estimated data.
What "Success" Actually Means for Ecosystem Restoration
As researchers evaluated their three strategies, a philosophical question emerged: what counts as successful restoration? Does a tree need to be genetically identical to the original American chestnut? Does it need to fill the exact same ecological role? Does it need to grow at the same rate and reach the same size?
These questions matter because they determine which strategy makes sense. If purity of genetics is paramount, pure breeding or perhaps genetic modification (which doesn't introduce foreign species genetics) wins. If speed matters, genetic modification or backcrossing is better. If the goal is simply getting a robust, disease-resistant tree back into the forest that can participate in the ecosystem in a functional way, even if it's a hybrid, then hybrids become viable.
The American Chestnut Foundation has been relatively pragmatic. The organization's position is that the ideal chestnut for restoration would be one that combines maximum fungal resistance with growth rates close to historical American chestnuts. If that combination requires some genetic modification or some hybrid genetics, so be it. The goal is ecosystem restoration, not creating a museum piece.
But not everyone agrees with that pragmatism. Some ecologists worry about releasing genetically modified trees into natural ecosystems. The long-term consequences of modifying an organism's genome are hard to predict. A modification intended to improve resistance to fungal infection might have unforeseen side effects that don't manifest for decades. Some conservationists prefer the classical breeding approach, arguing that it's more proven and less likely to create unintended consequences.
These aren't technical disagreements. They're fundamentally about risk tolerance and what we value in restoration.

The Role of the Blight-Suppressing Virus
One pathway to restoration that researchers have pursued less intensively involves the natural virus that infects the blight fungus itself. Cryphonectria hypovirus 1 (CHV-1) is a virus that attacks the fungus, weakening or killing it. In regions where this virus is present, chestnut trees can survive alongside the blight because the virus keeps fungal populations suppressed.
This suggested a biological control approach: intentionally introduce the virus into areas where the blight fungus is present but the virus is absent. If successful, the virus could establish itself and keep the blight in check, allowing surviving chestnuts to thrive without any need for genetic modification or hybrid breeding.
Biological control appeals to purists because it's entirely natural—a virus against a fungus, no human genetics involved. But it's proven difficult to implement. The virus doesn't spread spontaneously through fungal populations the way you'd hope. It requires direct contact between infected fungal spores and uninfected ones. In some regions, this happens naturally. In others, the virus never seems to establish.
Researchers have experimented with introducing the virus manually, applying it to infected trees in controlled ways. Sometimes it works. Sometimes the virus fails to persist. The reasons aren't fully understood, but they probably involve complex interactions between virus, fungus, environment, and host tree genetics.
Biological control remains a promising complementary strategy. Even if genetic resistance is the primary pathway for restoration, having the suppressive virus present in some areas could be an additional safeguard. But it's not a complete solution on its own.

The Timeline Problem: Patience as a Scientific Constraint
Perhaps the most underappreciated challenge in chestnut restoration is simply waiting. American chestnuts take years to mature. A single breeding cycle takes a decade. A backcrossing program involves 5-10 such cycles, easily spanning 50-100 years from start to finish.
This means that anyone starting a chestnut restoration project today will not live to see its completion. The scientists conducting the American Chestnut Foundation's research were planning for outcomes they would never personally witness. Graduate students who started projects would still be waiting for results in their 60s.
This timeline problem creates practical challenges. Funding agencies want to see results on funding cycles measured in years or maybe decades, not generations. Scientists need career advancement, requiring publications and progress reports. Institutions shift priorities. Public attention wanes. The knowledge accumulated by one generation of researchers might be lost before the next generation needs it.
Yet chestnuts, and all trees really, operate on their own timeline. You can't speed up a tree's maturation much. You can provide ideal growing conditions, which helps marginally, but the fundamental biology of how fast trees grow is hard to change. This is why genetic engineering appeals to some researchers—it offers the possibility of faster progress by circumventing slow breeding cycles.
But genetic engineering has its own long timeline for validation. Even if you create a transgenic tree, you need to test it in real-world conditions to make sure it actually works as predicted. You need to confirm that the beneficial traits (fungal resistance) persist across generations and that unexpected side effects don't emerge. This still requires decades of observation.

Different Restoration Strategies for Different Regions
One insight that emerged from the comprehensive testing was that different regions of the chestnut's historical range might benefit from different restoration strategies. Not all of Appalachia is identical. Elevation, latitude, humidity, and temperature vary significantly.
In cooler, drier mountain regions where the blight fungus grows more slowly, even modest resistance might be enough. A tree with a 60 or 70 on the 0-to-100 damage scale might survive well in these conditions. This means that pure-bred American chestnuts with naturally high resistance might be sufficient. Such a strategy would preserve genetic purity and would start with trees that already exist (the LSACs and their descendants).
In warmer, more humid lowland areas where the blight fungus grows aggressively, you'd need higher resistance. A tree with a 70 on the damage scale might still die in these conditions. You'd need hybrid genetics with maximum resistance, or transgenic modifications, or both. The trade-off of slower growth might be acceptable if it meant the tree could actually survive to reproduction.
In intermediate regions, different strategies might be optimal. The diversity of conditions across the chestnut's historical range means that a one-size-fits-all restoration approach is probably not realistic. Different regions might end up with different types of restored chestnuts.
This actually mirrors what happened historically. Before the blight, the American chestnut wasn't perfectly uniform across its range. Northern populations and southern populations had slight genetic differences, adapted to their respective climates. Restoration might recreate something similar—diverse chestnut populations adapted to their specific regions.

Real-World Testing and Adaptive Management
None of the three restoration strategies has been fully validated in the wild yet. All the testing has been done in nursery conditions or controlled experiments. A tree that performs well in a research nursery might fail in a real forest where it faces competition, deer browse, drought, disease pressure from other pathogens, and all the other stresses wild plants encounter.
This is why the next phase of chestnut restoration will likely involve careful, small-scale reintroductions. Plant restored chestnuts in protected areas where they can be monitored intensively. Track their survival, growth, reproduction, and health over decades. If they perform well, gradually expand the reintroduction. If problems emerge, adjust the approach.
This adaptive management approach is standard in restoration ecology, but it requires patience and humility. It means accepting that early attempts might fail. It means being willing to change course based on evidence. It means treating restoration as an ongoing experiment rather than a one-time intervention.
Some seed orchards are already growing pure-bred chestnuts for potential reintroduction. Hybrid seed orchards are producing backcrossed trees at various generations. Transgenic chestnuts are being cultivated in secure facilities, pending regulatory approval for field testing. Small-scale plantings have begun in states like New York and Pennsylvania, with close monitoring.
The earliest reintroductions might show results within 10-20 years, but full ecosystem restoration—actually restoring chestnuts to ecological dominance in Appalachian forests—would take centuries. You're talking about a multi-generational project that will outlive any individual scientist or institution.

The Regulatory and Philosophical Hurdles
Even if researchers solve all the biological problems, significant regulatory and philosophical hurdles remain. Genetic modification in particular faces skepticism from both regulatory agencies and the public. The FDA, USDA, and EPA all have authority over genetically modified plants, and approval processes can take years.
Beyond regulatory approval, there's the question of public acceptance. Would people want genetically modified chestnuts planted in public forests? Would they worry about unintended ecological consequences? Would they see it as playing God with nature?
Hybrid restoration faces less regulatory scrutiny—hybrids created through traditional breeding are not subject to the same approval processes as transgenics. But hybrids face their own philosophical objection: if the tree is part Chinese chestnut, is it really restoring the American chestnut or creating something new?
Pure breeding faces the fewest regulatory hurdles and the most acceptance from purists. But it's also the slowest approach and the one most uncertain to succeed. If the natural resistance available in surviving American chestnuts turns out to be insufficient, the entire approach fails.
Most likely, restoration will proceed with multiple strategies being pursued in parallel, with different regions potentially using different approaches based on local conditions and local preferences. Some areas might embrace genetic modification if it seems necessary. Others might insist on pure breeding or at most hybrid approaches. The result would be a mosaic of restoration efforts rather than a unified program.

International Implications and Future Applications
While this article focuses on American chestnuts, the work has implications for other species facing similar threats. Ash trees in North America are currently being decimated by an invasive beetle from Asia. European elms are threatened by an invasive fungus. These problems are global—climate change and global trade mean that pathogens spread constantly.
The American chestnut restoration project is essentially a proof of concept. If these approaches work for chestnuts, they could be adapted for other species. Genetic engineering approaches, for instance, could be developed for ash trees or elms. Backcrossing programs using disease-resistant relatives could be established. Biological control approaches using natural enemies of invasive pathogens could be deployed.
The techniques and knowledge developed during chestnut restoration will be valuable far beyond chestnuts. As invasive pathogens continue to threaten tree species globally, having multiple restoration strategies ready to deploy will be essential.

FAQ
What exactly happened to the American chestnut?
The American chestnut was nearly wiped out by two invasive fungal pathogens from Asia that arrived in the early 1900s. An estimated 3 billion trees died from chestnut blight and root rot disease between 1904 and 1950, making the species functionally extinct in its native Appalachian range. The fungus, Cryphonectria parasitica, spread rapidly and killed trees faster than they could reproduce, though the species wasn't completely extinguished—surviving populations persisted in western North America and botanical gardens.
How are scientists trying to restore the American chestnut?
Scientists are pursuing three main restoration strategies simultaneously. First, they're breeding naturally resistant American chestnuts together, selecting for the most disease-resistant individuals to hopefully concentrate genetic resistance over generations. Second, they're creating hybrid chestnuts by crossing American chestnuts with disease-resistant Chinese chestnuts and then backcrossing the hybrids with American genetics to increase American genetic content while maintaining Chinese resistance genes. Third, they're using genetic engineering to insert genes that help chestnuts fight the fungal pathogen, though this approach faces regulatory and growth-rate challenges.
Why does growth rate matter so much for restoration?
Growth rate matters because the historical dominance of American chestnuts in eastern forests was partly due to their rapid growth, which allowed them to compete aggressively for canopy space. Any restoration approach that produces slower-growing trees might create chestnuts that survive fungal infection but fail to regain ecological dominance in the forest. Researchers discovered that many disease-resistant trees grow more slowly, creating a biological trade-off between survival and growth performance.
How long will it take to restore American chestnuts to forests?
The timeline is measured in decades to centuries, depending on the strategy. A single breeding cycle in chestnuts takes 5-10 years since trees must mature before producing seeds. A complete backcrossing program might take 50-100 years. Genetic engineering requires decades of testing before field release. Even after trees are ready, establishing self-sustaining populations in wild forests could take multiple tree generations, potentially centuries. Full ecosystem restoration to historical chestnut dominance would take many centuries.
What are Large Surviving American Chestnuts (LSACs) and why are they important?
Large Surviving American Chestnuts are American chestnut trees that somehow survived exposure to chestnut blight in their native range, suggesting they possess some level of natural resistance. Scientists study LSACs to understand what makes them resistant and to breed with them to potentially concentrate resistance genes. However, testing showed that most LSACs have only modest resistance levels, suggesting natural resistance alone may be insufficient without additional breeding or genetic approaches.
What's the difference between hybrid and pure-bred restoration approaches?
Pure-bred restoration uses only American chestnut genetics, attempting to breed together the most naturally resistant American chestnuts to concentrate resistance while maintaining genetic purity. Hybrid restoration crosses American chestnuts with disease-resistant Chinese chestnuts to gain proven resistance genes, then backcrosses repeatedly to increase American genetic content. Pure breeding preserves genetic identity but is slow and depends on limited natural resistance. Hybrid approaches are faster but introduce non-American genetics, and hybrids often grow more slowly than pure American chestnuts.
What is the oxalic acid degradation approach in genetic engineering?
Researchers identified oxalic acid as a key factor in how the blight fungus damages chestnut trees. They inserted a gene from wheat that produces an enzyme capable of breaking down oxalic acid into the American chestnut genome. The theory was that chestnuts with this added ability could neutralize one of the fungus's main weapons. However, the inserted gene unfortunately disrupted a growth-controlling gene in chestnuts, causing most transgenic trees to grow significantly slower than normal, though a small percentage showed both good resistance and normal growth.
Why haven't genetically modified chestnuts been released into forests yet?
Genetically modified organisms require regulatory approval from multiple agencies including the FDA, USDA, and EPA before field release. The approval process can take years and requires extensive testing to demonstrate safety. Beyond regulatory requirements, there are public concerns about releasing genetically modified organisms into natural ecosystems, and uncertainty about long-term ecological consequences. Additionally, researchers are still optimizing the genetic modification approach since the current version causes unexpected growth penalties.
Could biological control using blight-suppressing viruses be a solution?
A naturally occurring virus called Cryphonectria hypovirus 1 (CHV-1) infects and weakens the blight fungus, allowing chestnuts to survive in areas where the virus is present. Some researchers have explored intentionally introducing this virus to areas where the blight exists but the virus doesn't. However, the virus doesn't spread spontaneously the way hoped, and establishing it in new regions has proven difficult and inconsistent. Biological control remains a promising complementary strategy but isn't sufficient as a standalone solution.
How do climate and regional conditions affect chestnut restoration strategies?
Different strategies may be optimal for different regions of the chestnut's historical range. In cooler, drier mountain regions where the blight fungus grows more slowly, even modest natural resistance might be sufficient, making pure breeding approaches viable. In warmer, more humid lowland regions where the fungus is more aggressive, higher levels of resistance are needed, requiring hybrids or genetic modification. This suggests that successful restoration might involve different strategies adapted to regional conditions rather than one universal approach.
What happens to restored chestnuts if a new pathogenic threat emerges?
One concern with any restoration strategy is that while focused on solving the blight problem, restored chestnuts might become vulnerable to different pathogens. This is why restoration programs involve monitoring for other diseases and maintaining diverse genetic backgrounds rather than creating a single "super-chestnut." A population with genetic diversity would be more likely to have some individuals able to withstand novel disease threats, similar to how the original pre-blight chestnut population had variation.
The Future of American Chestnut Restoration
The American Chestnut Foundation's comprehensive study published in 2026 represents a pivotal moment in restoration science. For the first time, researchers had systematically compared the three main restoration strategies using rigorous, quantifiable methods. The findings weren't simple—none of the strategies emerged as clearly superior to the others. Instead, each strategy offered different advantages and trade-offs.
Pure breeding preserved genetic authenticity but was slow and uncertain. Backcrossing with Chinese chestnuts promised faster progress but introduced foreign genetics and growth penalties. Genetic engineering could theoretically be fastest but faced its own complications around gene insertion and growth costs.
The most likely scenario is that restoration proceeds with all three strategies in parallel, pursued by different researchers and organizations, with results from each approach informing the others. Success will probably come from whichever strategy proves most robust across the longest timeline and most likely to produce trees that survive, grow, and reproduce in real forest conditions.
What makes this work scientifically remarkable is its scale of ambition and patience. Scientists are committing themselves and their institutions to a project whose results they may never personally see. They're working on a timeline measured in centuries. They're accepting that early efforts might fail and that adjustment based on evidence is essential.
In a landscape of rapid technological change and quick results, chestnut restoration represents something different: careful, methodical work guided by evidence and constrained by the fundamental biology of living organisms. It's a reminder that some problems require patience, and that bringing back what we've lost isn't a matter of clever engineering so much as careful understanding of how nature works.
The American chestnut didn't disappear because of any failure of the tree itself. It lost a biological arms race against an invasive fungus it had never encountered before. Restoration isn't about making the tree stronger in some abstract sense. It's about equipping it with the specific defenses needed to survive in a world that includes pathogens that evolved over millions of years on the opposite side of the planet.
That project—matching evolutionary innovation against evolutionary innovation, using classical breeding, genetic engineering, and adaptive management—might actually be the truest form of ecosystem restoration available. It's not about returning forests to some imagined pristine past. It's about understanding the biological forces that shape forests and working deliberately with those forces to recreate something valuable that we lost.
The chestnuts sprouting from root systems in Appalachian forests today don't know what restoration means. They're just trying to grow and survive, the way their ancestors did for millions of years. Whether genetic engineering, hybrid breeding, or classical selection ultimately succeeds, the goal is the same: to give those sprouts a fighting chance at what their ancestors took for granted.

Key Takeaways
- American chestnuts were functionally extinct after 3 billion trees died from invasive Asian fungi within 50 years of arrival in 1904
- Three restoration strategies are being tested: pure breeding of naturally resistant American chestnuts, backcrossing with disease-resistant Chinese chestnuts, and genetic engineering to enhance fungal resistance
- Over 14 years, researchers tested more than 4,000 individual trees, finding most American chestnuts have only modest natural resistance to blight
- All restoration approaches face a growth-rate trade-off where disease resistance often comes paired with slower growth than historical chestnuts
- Restoration timelines span 50-100+ years per strategy, making this a multi-generational project requiring institutional and scientific patience
![Restoring the American Chestnut: Modern Genetics Brings Back a Forest Icon [2025]](https://tryrunable.com/blog/restoring-the-american-chestnut-modern-genetics-brings-back-/image-1-1770925354415.jpg)



