The WHO's Unprecedented Condemnation of a US Vaccine Trial
In February 2026, the World Health Organization took the extraordinary step of publicly denouncing a US-funded vaccine trial as fundamentally unethical. This wasn't a quiet disagreement buried in academic journals. The WHO issued a formal statement, systematically dismantling the trial's scientific and ethical foundations point by point. The trial in question would withhold hepatitis B vaccine birth doses from thousands of newborns in Guinea-Bissau, Africa, a country where over 12 percent of adults carry chronic hepatitis B.
The controversy centers on research conducted by Danish scientists Christine Stabell Benn and Peter Aaby of the Bandim Health Project at the University of Southern Denmark. The Centers for Disease Control and Prevention, under the leadership of Health Secretary Robert F. Kennedy Jr., awarded them $1.6 million for this work. The funding was announced through a Federal Register notice in December 2025, sparking immediate backlash from global health experts who questioned both the science and the ethics of the proposed study.
What makes this situation particularly striking is its backdrop. Kennedy has been a vocal critic of vaccine programs and has used controversial research from Benn's team to justify cutting global vaccine funding. Meanwhile, the CDC's own advisory committee, staffed with Kennedy appointees, recently abandoned the longstanding universal recommendation for hepatitis B vaccine birth doses. Against this political context, the new funding raised red flags that extended far beyond the scientific community.
The trial represents something increasingly rare in modern medicine: a proposal to deliberately withhold a proven, established, lifesaving intervention from study participants for research purposes. This crosses ethical lines that international research standards were specifically designed to prevent. Understanding why the WHO reacted so forcefully requires examining the science, the history, and the broader implications for how vaccine research is conducted globally.
TL; DR
- The Core Issue: The CDC funded a trial that would withhold hepatitis B vaccine birth doses from 14,000 newborns in Guinea-Bissau to study alleged safety concerns
- WHO Position: The organization formally condemned the trial as unethical, lacking scientific justification, and potentially exposing infants to serious harm
- Political Context: Robert F. Kennedy Jr.'s leadership at HHS coincided with both the trial funding and removal of hepatitis B birth dose recommendations
- Global Standard: Hepatitis B birth dose vaccination is used by 115+ countries and prevents mother-to-child transmission, liver disease, and cancer
- Current Status: The trial remains suspended pending ethical and technical review in Guinea-Bissau, though US health officials claim it's "proceeding as planned"
Benn and Aaby's research has significant influence among vaccine skeptics and in media coverage, but less so in the scientific community and public health policy. Estimated data based on narrative context.
Understanding Hepatitis B and Why Birth Doses Matter
Hepatitis B is far more serious than many people realize. The virus attacks the liver and can cause chronic infection, cirrhosis, liver cancer, and death. What makes hepatitis B particularly dangerous is how efficiently it transmits from mother to child at birth. Without intervention, infected mothers pass the virus to their babies up to 90 percent of the time.
The hepatitis B vaccine birth dose represents one of modern public health's most elegant solutions to this problem. A single dose given within 24 hours of birth interrupts mother-to-child transmission with remarkable effectiveness. The intervention has been used globally for over three decades. More than 115 countries now include it in their national vaccination schedules. The evidence supporting this practice is overwhelming and built on decades of real-world experience.
Guinea-Bissau currently has a particularly high burden of hepatitis B. Over 12 percent of the adult population carries chronic infection. This makes the birth dose vaccination especially important for preventing disease in the next generation. The country had already decided to implement hepatitis B birth dose vaccination in 2028 as part of its public health improvements. The only reason for the delayed timeline was resource constraints, not scientific doubt about the vaccine's value.
The Historical Success of Hepatitis B Vaccination Programs
Before hepatitis B vaccines existed, the virus killed hundreds of thousands of people annually. Countries that implemented universal infant hepatitis B vaccination saw dramatic declines in chronic infection rates. Hong Kong, for example, reduced hepatitis B surface antigen prevalence from over 13 percent in vaccinated cohorts to less than 1 percent within a generation. These weren't subtle improvements—they were transformative public health achievements.
The birth dose specifically targets the window when transmission risk is highest. Vaccinating at six weeks, as Guinea-Bissau currently does, misses the critical neonatal period when mother-to-child transmission most often occurs. Infants born to infected mothers who don't receive birth dose protection face a narrow window of vulnerability. Moving vaccination to birth closes that gap entirely.
Why Guinea-Bissau's Context Matters
Guinea-Bissau isn't an arbitrary choice for this trial. The country has limited healthcare resources, which makes it an attractive location for research involving delayed interventions. However, this same resource constraint creates an inherent ethical problem. Researchers can exploit scarcity by studying populations that lack resources for interventions that wealthier countries take for granted. The WHO explicitly called this out: "Exploiting scarcity is not ethical."
This dynamic has deep historical roots. Colonial-era medical research often used vulnerable populations in Africa to test interventions without their meaningful consent. While standards have theoretically improved, the underlying power imbalance persists. A trial that would never be approved in the United States or Europe becomes possible in a resource-limited country, particularly when funded by a wealthy nation's health agency.
The birth dose of hepatitis B vaccination is estimated to have a higher safety score compared to the six weeks dose, highlighting the ethical concerns of withholding the proven intervention. Estimated data.
Who Are Benn and Aaby? The Controversial Research Team
Christine Stabell Benn and Peter Aaby have become controversial figures in vaccine safety research. Their work has consistently questioned the safety profiles of established vaccines, particularly focusing on claims about unintended harm from routine immunizations. While scientific debate is healthy, the medical community has raised significant concerns about their methodology, conclusions, and influence.
Their research has generated hypotheses that contradict data from large, well-controlled studies conducted globally. For example, they've suggested that certain vaccines might increase mortality from other causes, a claim not supported by extensive epidemiological evidence. Yet their work has gained outsized influence in certain circles, particularly among vaccine skeptics and in media coverage critical of vaccination programs.
The Track Record of Benn and Aaby's Research
One of their most notable works studied what they called "non-specific effects" of vaccines, suggesting that some vaccines might protect against diseases they weren't designed for, while others might increase susceptibility. While the theory was intriguing, subsequent research by other groups failed to replicate their findings with rigor. Independent analysis suggested their studies had methodological limitations that made their conclusions unreliable.
Despite these concerns, their research has been cited by prominent vaccine skeptics, including Robert F. Kennedy Jr. Kennedy has explicitly referenced their work when making public statements about vaccine safety. This connection between the research team and Kennedy's policy positions creates an apparent conflict of interest that undermines the credibility of both the science and the funding decision.
Why This Matters for Research Ethics
The selection of Benn's team for this particular trial raises questions about funding allocation. The CDC gave them $1.6 million through a "non-competitive, unsolicited proposal" process. This means researchers approached the agency with their idea, and the CDC awarded funding without a competitive application process. Such arrangements are legitimate in principle but are typically reserved for exceptionally strong proposals from established teams.
However, Benn's team's track record didn't justify extraordinary trust in their judgment about what constitutes ethical research. Their previous work suggested they were willing to design studies that challenged established safety data. Funding them to conduct a trial that withheld a proven vaccine created the appearance that funding decisions were influenced by Kennedy's known views about vaccines.
The CDC's Shift on Hepatitis B Birth Dose Recommendations
The timeline of events is telling. In 2025, the CDC's advisory committee voted to remove the universal recommendation for hepatitis B vaccine birth doses. Instead of recommending all newborns receive the vaccine at birth, the new guidance suggested it for infants born to mothers with known hepatitis B infection. This was a significant policy change from the longstanding recommendation for universal birth dose vaccination.
The advisory committee that made this decision consisted of advisors handpicked by Robert F. Kennedy Jr. This timing—coupled with the funding announcement for Benn's trial just months later—suggested a coordinated shift in vaccine policy toward less aggressive immunization schedules. Global health experts widely condemned the decision to remove the birth dose recommendation, arguing it would leave millions of infants unprotected against a serious disease.
The Rationale for Removing Universal Recommendations
The CDC's new guidance claimed that selective vaccination—only vaccinating infants born to infected mothers—would be safer and more efficient. However, this logic ignores a fundamental problem: many mothers with hepatitis B are unaware of their infection status. Testing all pregnant women for hepatitis B isn't done universally in many countries, including resource-limited settings like Guinea-Bissau. By restricting vaccination to known cases, the policy would leave unidentified cases unprotected.
Universal birth dose vaccination avoids this problem entirely. Every newborn gets protected regardless of the mother's status. This approach has been proven effective globally for decades. The shift to selective vaccination represented a departure from the successful strategy that had reduced hepatitis B transmission worldwide.
The Political Context of the Policy Change
Robert F. Kennedy Jr. became a prominent vaccine skeptic over several decades. He has consistently questioned the safety and necessity of routine childhood vaccines. When he was appointed to lead the Department of Health and Human Services in 2025, concerns about vaccine policy immediately surfaced. His appointment gave skeptics influence over vaccine recommendations that had been established by scientific consensus.
The timing of these policy changes couldn't have been more problematic. The CDC advisory committee reconsidered hepatitis B birth dose recommendations and voted to make them less universal. Simultaneously, funding flowed to researchers known for questioning vaccine safety. These weren't isolated decisions—they reflected a coherent shift in vaccine policy influenced by Kennedy's skepticism.
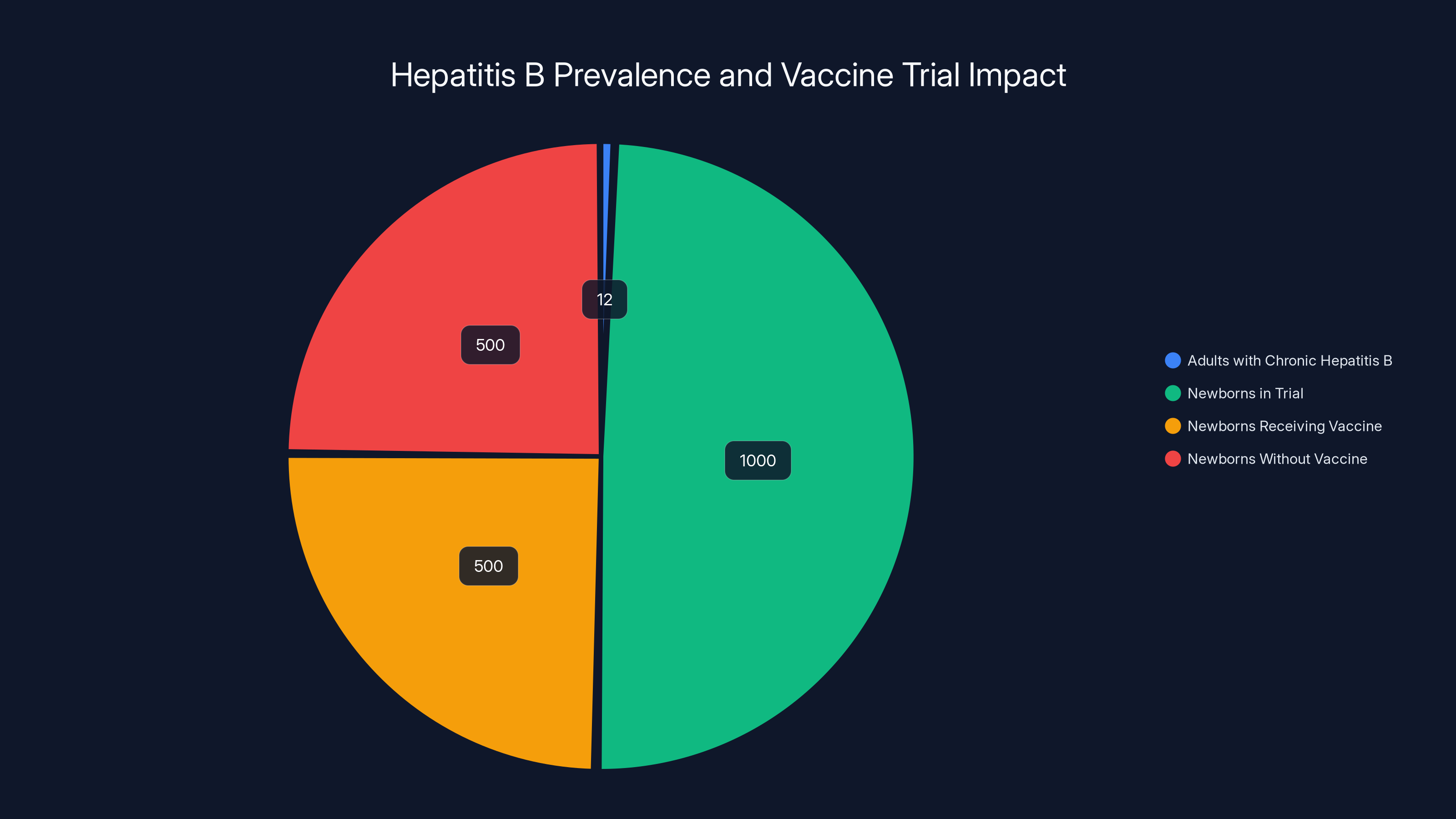
Estimated data shows the impact of withholding hepatitis B vaccines from newborns in a trial, highlighting ethical concerns. Estimated data.
The Trial Design: Withholding Proven Intervention for Research
The proposed trial would randomize 14,000 newborns in Guinea-Bissau into two groups. One group would receive hepatitis B vaccination at birth, as recommended by global health standards. The other would receive their first dose at six weeks, following Guinea-Bissau's current practice. Researchers would then track both groups for alleged safety differences.
On its surface, this might sound like a reasonable comparison of two vaccination schedules. The reality is far more ethically problematic. One schedule represents the proven, evidence-based standard. The other represents the current practice only because of resource constraints, not scientific preference. The trial would essentially compare the standard of care in wealthy countries against a suboptimal practice in a poor country.
The Fundamental Ethical Problem
With a proven intervention available, deliberately withholding it for research purposes requires extraordinary justification. International ethical guidelines like the Declaration of Helsinki require that research protocols not deviate from the best proven methods without compelling reason. There must be no credible evidence that the withheld intervention causes harm. There must be scientific uncertainty about which approach is better.
None of these conditions were met in this trial. The hepatitis B birth dose is proven effective. There is no credible evidence that vaccinating at birth causes harm compared to vaccinating at six weeks. There is no genuine scientific uncertainty about whether birth dose vaccination prevents mother-to-child transmission. From an ethical standpoint, the trial was unjustifiable.
The Methodological Problems
Beyond the ethical issues, the trial had severe methodological limitations. It was designed as a single-blind, no-treatment-controlled study. Single-blind design means only participants are unaware of their assignment, not researchers. This creates significant risk of bias. Researchers knowing who received which schedule could unconsciously influence how they assess outcomes.
Moreover, a "no-treatment control" doesn't make sense for a vaccine trial. The comparison should be between two active interventions at different times. Instead, the trial essentially compared getting vaccinated at birth against getting vaccinated later. The design would make it difficult to determine what actually caused any observed differences.

The WHO's Detailed Ethical Critique
The World Health Organization's statement wasn't vague criticism. The organization provided a systematic, point-by-point analysis of why the trial violated established ethical and scientific principles. This level of detailed, public condemnation from WHO is unusual, indicating the severity of the ethical violations.
WHO highlighted that the trial protocol "does not appear to ensure even a minimum level of harm reduction and benefit to study participants." The organization specifically noted that withholding hepatitis B vaccination from newborns exposed to the virus at birth represented unnecessary harm. In research ethics, the principle of non-maleficence—doing no harm—is fundamental. This trial couldn't meet that basic standard.
WHO's Specific Ethical Concerns
First, the WHO noted that there was no credible scientific evidence justifying the hypothesis that birth dose vaccination causes harm. Benn's team had published concerns about alleged non-specific effects of vaccines, but these findings hadn't been replicated or validated by independent research teams. Basing a trial on unproven, contentious claims was methodologically unsound.
Second, the WHO emphasized that withholding an established, lifesaving intervention violates the ethical principle that research must offer reasonable expectation of benefit to participants or the population they represent. A trial that withheld proven protection against a serious disease to some participants offered no compensating benefit.
Third, the organization criticized the trial's design for creating substantial bias risk. Single-blind designs are considered inferior in vaccine research. The lack of an active control group—comparing birth dose to another protective approach—made results difficult to interpret. A biased, difficult-to-interpret study lacks scientific value.
Fourth, WHO noted that exploiting resource constraints to conduct research that wouldn't be approved in wealthy countries violates research ethics principles. Guinea-Bissau was chosen partly because the country hadn't yet implemented hepatitis B birth dose vaccination due to funding limitations. Using scarcity as a reason to conduct ethically questionable research is precisely what international guidelines are meant to prevent.
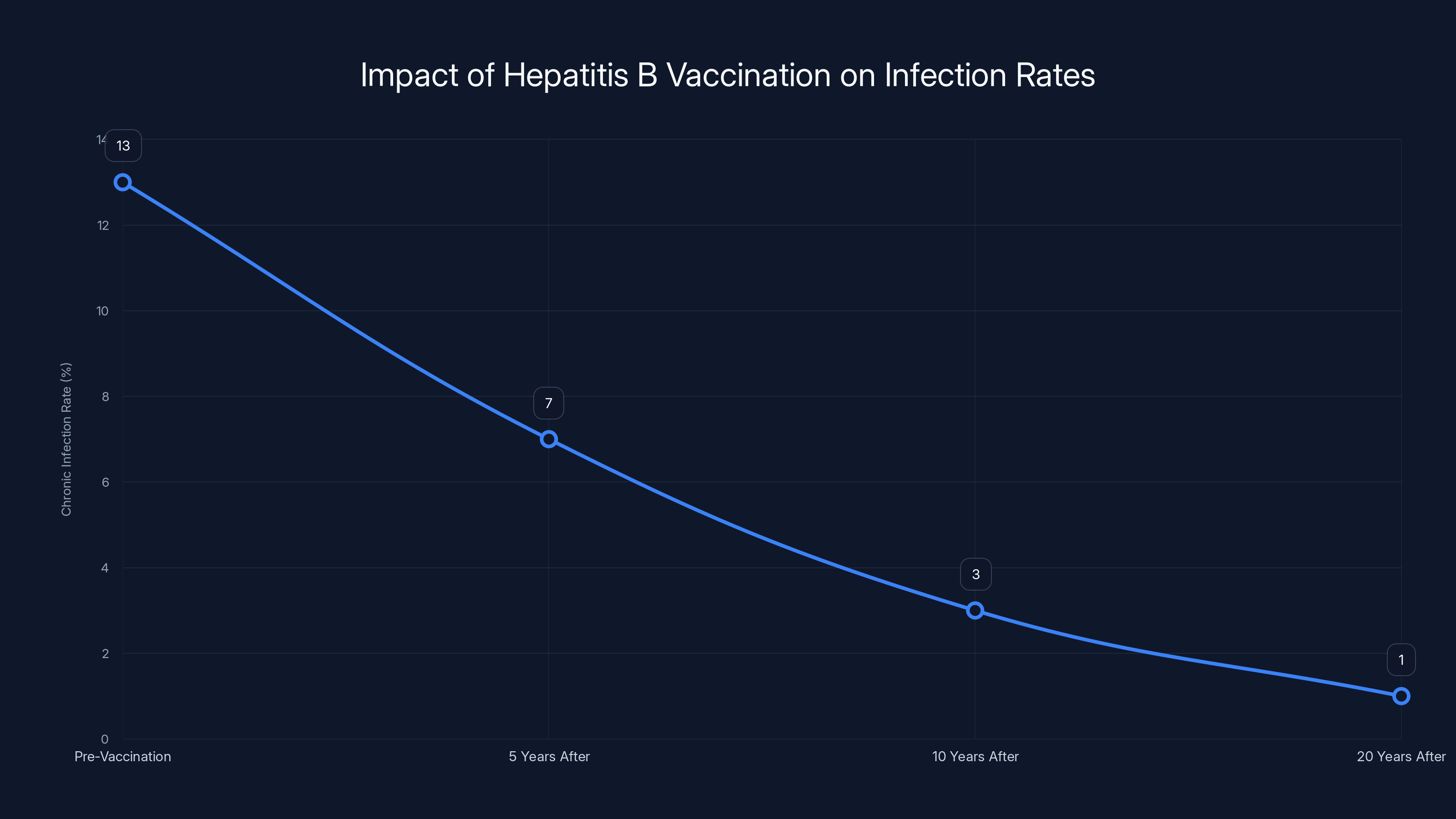
Estimated data shows a dramatic decline in chronic infection rates following the introduction of universal hepatitis B vaccination, highlighting its effectiveness in reducing transmission.
Global Standards for Hepatitis B Birth Dose Vaccination
Hepatitis B birth dose vaccination isn't some experimental intervention. It's been part of routine immunization schedules in wealthy countries for decades. The United States recommends it universally. Europe recommends it universally. Over 115 countries have adopted hepatitis B birth dose vaccination as standard practice.
This global consensus emerged because the evidence is overwhelming. Birth dose vaccination prevents mother-to-child transmission in over 95 percent of cases. It prevents chronic infection in infants who would otherwise be infected. It prevents liver cancer, cirrhosis, and death decades later. The long-term benefit of universal birth dose vaccination programs is beyond dispute.
Why Guinea-Bissau Chose to Implement Birth Dose
Guinea-Bissau's government didn't hesitate to decide on hepatitis B birth dose implementation. They planned to adopt it in 2028 specifically because of its proven benefits. The only delay was practical—securing resources to implement the program. There was no debate about whether birth dose vaccination would help. Everyone agreed it would.
This context makes the trial's purpose unclear. If the government was already committed to implementation, why conduct a trial comparing birth dose to delayed vaccination? The research wouldn't change policy—the policy was already decided. The trial seemed designed to generate data questioning birth dose benefits, regardless of what evidence actually showed.
The Mother-to-Child Transmission Problem
In Guinea-Bissau, hepatitis B mother-to-child transmission is a significant cause of chronic infection. Women with hepatitis B who deliver babies face a critical window where infection transmission is most likely. Vaccinating newborns at birth closes that window.
Delaying vaccination to six weeks leaves newborns vulnerable for 42 days. This gap is particularly significant for infants born to chronically infected mothers. Even with other protective measures like hepatitis B immunoglobulin, birth dose vaccination substantially reduces transmission risk. Deliberately extending this vulnerable period for research purposes exposes infants to unnecessary risk.

The Role of Robert F. Kennedy Jr. in Vaccine Policy
Robert F. Kennedy Jr.'s influence on this situation extends far beyond the trial funding. His appointment as Health Secretary represented a fundamental shift in US vaccine policy direction. Kennedy has spent decades questioning vaccine safety and promoting theories that contradict scientific evidence. His government positions gave those views direct influence over federal health agencies.
Kennedy has explicitly cited research by Benn and Aaby when making public statements about vaccine safety concerns. He has claimed that vaccines cause various conditions without evidence. He has promoted conspiracy theories about vaccine development. Placing such a person in charge of health agencies raised immediate concerns about vaccine policy changes.
Kennedy's Track Record on Vaccines
Kennedy founded an organization called the Children's Health Defense, which promotes vaccine skepticism. The organization has spread vaccine misinformation extensively. Kennedy himself has claimed that vaccines cause autism, despite the original autism study being fraudulent and retracted. He has suggested that vaccines are more dangerous than the diseases they prevent, contradicting epidemiological evidence.
Yet Kennedy succeeded in gaining influence over vaccine policy. His leadership at HHS gave him direct control over the CDC and other health agencies. Within months, vaccine recommendations changed. Funding flowed to controversial researchers. Global health programs faced cuts. Kennedy's skepticism directly influenced policy outcomes.
The Conflict of Interest Problem
Funding a trial conducted by researchers whose work Kennedy has cited creates an obvious conflict of interest. It appears that funding decisions were influenced by ideology rather than scientific merit. The non-competitive nature of the funding award makes this worse. If the CDC had opened this to competitive bidding, other research teams might have submitted superior proposals.
The timing adds another layer of concern. The trial was funded months after the CDC advisory committee, staffed with Kennedy appointees, voted to remove hepatitis B birth dose recommendations. This sequential decision-making suggests coordination—first change policy recommendations, then fund research that questions the old policy.

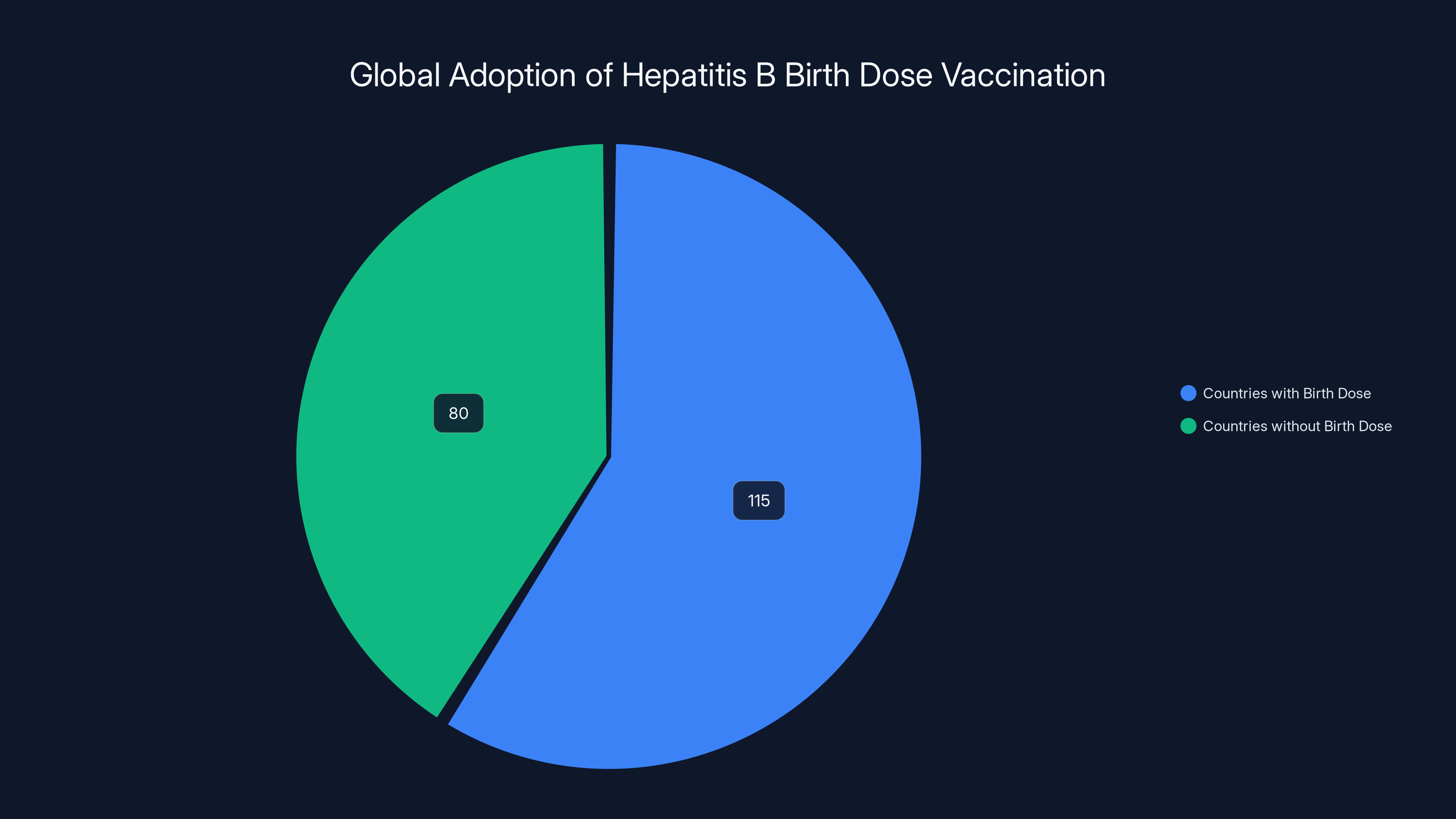
Out of approximately 195 countries, over 115 have adopted hepatitis B birth dose vaccination, highlighting its global acceptance and importance in preventing mother-to-child transmission. Estimated data.
International Research Ethics Standards
The field of research ethics emerged partly in response to historical abuses. The Tuskegee syphilis study, Nazi medical experiments, and Japanese biological warfare research demonstrated the dangers of unrestricted human subjects research. International standards were developed to prevent such abuses.
The Declaration of Helsinki is the primary ethical framework for medical research involving human subjects. It requires that research protocols provide a reasonable expectation of benefit or don't deviate unjustifiably from standard care. Research on vulnerable populations requires additional protections. Withholding proven interventions requires extraordinary justification. These aren't suggestions—they're fundamental ethical requirements.
Core Principles Violated by the Trial
The trial violated several core principles. Beneficence—the principle of providing benefit to research participants—wasn't met. Withholding proven protection doesn't benefit participants. Justice—the principle that research burdens shouldn't fall disproportionately on vulnerable populations—wasn't met. A trial that would never be approved in wealthy countries was proposed for a poor country.
Non-maleficence—the principle of avoiding harm—clearly wasn't met. Withholding hepatitis B vaccination from infants exposes them to serious harm. Informed consent requirements also weren't met. Participants in Guinea-Bissau couldn't meaningfully consent when the research offered them suboptimal care compared to global standards.
Why These Standards Matter
Research ethics standards exist because history demonstrates that researchers, even well-meaning ones, can convince themselves that ends justify means. Standards provide objective guidelines that prevent researchers' judgment from becoming corrupted by ideology or financial incentives. Violating these standards doesn't just harm current study participants—it undermines the entire system that protects vulnerable populations.
When wealthy countries conduct trials in poor countries that wouldn't be approved at home, it perpetuates colonial patterns. Research happens on vulnerable populations rather than with them. Findings that wouldn't be scientifically defensible become possible because of resource constraints. This is precisely what international ethics standards are meant to prevent.
The Current Status: Suspension and Uncertainty
As of early 2026, the trial's status remained unclear. Guinea-Bissau's minister of public health, Quinhin Nantote, announced that the trial was suspended pending technical and ethical review. The government had received insufficient coordination and information to make a final decision about approval. They chose the cautious path of pausing the trial until concerns could be addressed.
However, the US Department of Health and Human Services provided a statement claiming the trial was "proceeding as planned." This contradiction created confusion about whether the trial would actually move forward. Meanwhile, the Africa Centres for Disease Control and Prevention suggested the trial wouldn't proceed, aligning with Guinea-Bissau's suspension decision.
What the Suspension Means
Guinea-Bissau's suspension was significant. It meant the host country government exercised its authority to protect its population from ethically questionable research. This is how the system is supposed to work—host countries should maintain sovereignty over research conducted on their populations. When ethical concerns arise, countries should be able to say no.
The suspension also created practical difficulties for the trial team. Conducting research in a country without government approval is impossible in modern contexts. They can't access participants, facilities, or collaborating institutions without government support. Even if the US side claimed the trial was proceeding, it couldn't actually happen without Guinea-Bissau's participation.
The Broader Implications
This situation set a precedent. Other countries considering vaccine research might now demand higher ethical standards. They might refuse to host trials that would raise concerns in wealthy nations. This could slow research on legitimate questions, but it also ensures that vulnerable populations aren't used to test substandard interventions.
The trial's suspension also demonstrated that ethical concerns about US-funded research could overcome political pressure. Guinea-Bissau's government prioritized protecting its population despite funding incentives. This shows that established ethical principles still have force, even when challenged by powerful actors.

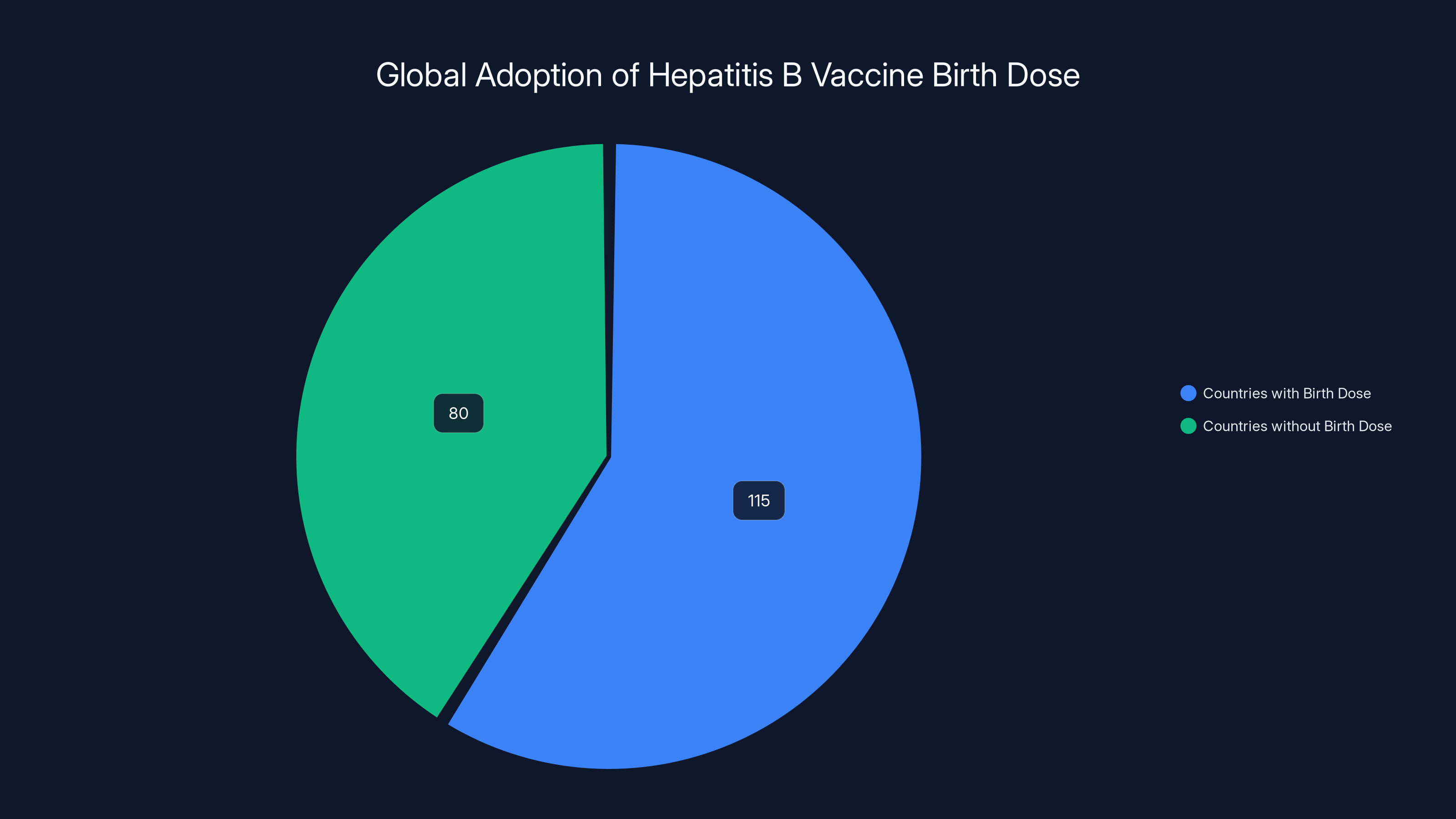
An estimated 115 countries have adopted the hepatitis B vaccine birth dose as a standard practice, highlighting its global acceptance and importance in preventing hepatitis B transmission.
Why This Matters Beyond Hepatitis B
This controversy isn't really about hepatitis B specifically. It's about research integrity, power dynamics in global health, and whether wealthy countries will respect ethical standards when conducting research in poor countries. The hepatitis B vaccine is just the vehicle through which these larger issues manifest.
The trial represents a broader pattern of concerning vaccine policy changes. Within months of Kennedy taking office, vaccine recommendations were relaxed. Vaccine program funding faced cuts. International health initiatives were reduced. Research funding flowed to teams known for vaccine skepticism. These weren't isolated decisions—they reflected a systematic shift toward less robust vaccine programs.
The Impact on Vaccine-Preventable Diseases
If vaccine programs are weakened globally, the consequences could be severe. Diseases that have been controlled in many countries could resurge. Measles, polio, diphtheria, and other serious illnesses that kill thousands annually could increase. The progress made over decades through vaccination could be lost.
Hepatitis B specifically affects hundreds of millions of people. A single prevention failure in a single country wouldn't create a major outbreak. But sustained weakening of hepatitis B prevention programs globally could allow chronic infection rates to rise. Over years and decades, this translates to increased cancer, cirrhosis, and liver disease globally.
The Precedent for Research Standards
Beyond disease-specific concerns, allowing this trial to proceed would establish a troubling precedent. It would suggest that withholding proven interventions is acceptable in research if conducted in low-income countries. It would suggest that controversial researchers can access federal funding despite questionable track records. It would suggest that research ethics standards are advisory rather than binding.
These precedents have long-term implications for all research conducted in vulnerable populations. If this trial is approved and proceeds, other researchers will reference it. They'll argue that their own ethically questionable proposals should also be funded. The standards protecting vulnerable populations globally become eroded.

Expert Consensus on the Trial's Ethical Status
Beyond the WHO, other prominent health experts have condemned the trial. Global immunization experts, pediatricians, and public health officials have stated clearly that the trial violates ethical standards. This isn't a controversial position within the medical community. There's broad, near-universal agreement that the trial shouldn't proceed.
The rare exceptions come from researchers closely aligned with Benn's team or with Kennedy's skeptical views about vaccines. Even vaccine skeptics sometimes acknowledge that withholding proven interventions goes too far. The trial sits at the extreme end of problematic vaccine research, not at some middle ground of legitimate debate.
Medical Societies' Positions
The American Academy of Pediatrics, the American Medical Association, and other medical societies have not endorsed this trial. They've remained largely silent, which in itself suggests disapproval. Medical organizations don't typically stay quiet about research they consider scientifically sound and ethically acceptable. Their silence indicates the trial is indefensible from a medical standpoint.
International pediatric organizations have similarly not endorsed the trial. This includes European pediatric societies, Asian pediatric organizations, and African health groups. Global pediatricians recognize that hepatitis B birth dose vaccination is standard, proven care that benefits children.

Looking Forward: What Happens Next
The trial's ultimate fate remains uncertain. If Guinea-Bissau maintains its suspension, the trial cannot proceed there. The research team could seek to conduct the trial in other countries, but they would face similar ethical scrutiny elsewhere. No major country is likely to approve a trial that withhold proven hepatitis B vaccination from newborns.
Alternatively, the trial could be redesigned to address ethical concerns. Instead of comparing birth dose to no vaccination, it could compare birth dose timing strategies where all participants receive the vaccine. It could focus on implementation questions rather than trying to generate doubt about vaccine benefits. A redesigned version might be ethically acceptable, though it would be a fundamentally different study.
The Broader Policy Implications
Regardless of this specific trial's outcome, the fact that it was funded at all signals something troubling about current vaccine policy in the US. The CDC funded it despite the ethical concerns. The advisory committee changes preceded it. These decisions suggest that skepticism about vaccines is now influencing federal health policy.
This could lead to further policy changes affecting vaccine recommendations globally. If the US weakens its stance on vaccines, other countries might follow. International organizations like WHO maintain positions based on evidence, but US policy changes still influence global discussions. Weakening US commitment to evidence-based vaccine programs could have ripple effects worldwide.
A Path Back to Evidence-Based Policy
Reverting to evidence-based vaccine policy requires acknowledging that vaccines are among the most successful public health interventions ever developed. They've prevented millions of deaths and billions of cases of serious disease. New vaccine policies should be based on scientific evidence and public health data, not ideology or fringe theories.
The hepatitis B vaccine specifically has prevented countless cases of liver cancer, cirrhosis, and death. The birth dose prevents mother-to-child transmission. The evidence is overwhelming. Any policy changes regarding hepatitis B vaccination should be grounded in this evidence, not in controversial theories that haven't been validated by independent research.
Lessons for Research Integrity and Ethics Oversight
This situation offers important lessons for how research ethics should function. The system is designed with multiple checkpoints: institutional review boards, host country oversight, professional ethical standards, and international guidelines. In this case, multiple checkpoints caught the same problem simultaneously.
However, the trial funding proceeded despite these concerns. IRBs presumably approved the trial protocol. The funding was awarded. It took international pressure from the WHO to halt it. The lesson here is that ethical oversight requires both good-faith implementation and sufficient independence from political pressure.
Strengthening Research Ethics
One key lesson is that ethics committees need genuine independence. If they're controlled by officials who are ideologically skeptical of vaccines, they can't objectively evaluate vaccine trials. Ethics review must be insulated from political pressure and ideological goals. Reviewers should be selected for expertise and commitment to ethical standards, not for ideological alignment.
Another lesson is that transparency matters. The trial became controversial because funding was announced publicly. If it had been conducted quietly without public notice, it might have proceeded without review. Transparency enables oversight by the scientific community and general public. Researchers and funders should assume their work will be scrutinized.
The International Dimension
This situation also highlights the importance of international oversight. National governments sometimes pursue policies that contradict global health evidence. International organizations like WHO can provide counterweight. When national policy diverges sharply from international scientific consensus, having global oversight mechanisms becomes critical.
However, international oversight works best when combined with local oversight. Guinea-Bissau's government suspension of the trial was crucial. Host countries must maintain authority over research conducted on their populations. They must be able to say no when external research doesn't serve their population's interests, regardless of funding offered.

FAQ
What is the hepatitis B vaccine birth dose, and why is it important?
The hepatitis B vaccine birth dose is a single injection given within 24 hours of birth to prevent mother-to-child transmission of hepatitis B virus. It's crucial because hepatitis B can transmit from infected mothers to newborns during delivery, with up to 90 percent transmission efficiency if no preventive measures are taken. This vaccine prevents chronic infection, liver disease, cirrhosis, and liver cancer, protecting infants throughout their lives. Over 115 countries have adopted birth dose vaccination as standard practice because the evidence demonstrates its effectiveness unequivocally.
Why did the WHO condemn this particular vaccine trial?
The WHO condemned the trial because it would deliberately withhold a proven, established, lifesaving vaccine from thousands of newborns to study alleged safety concerns that lack scientific justification. International research ethics standards prohibit withholding known beneficial interventions without extraordinary reasons. Since no credible evidence suggests hepatitis B birth dose causes harm, and since the vaccine prevents serious disease, the trial violated fundamental ethical principles. The WHO further noted that the trial's design created significant bias risk, limiting scientific interpretability, and that it exploited resource constraints in Guinea-Bissau to conduct research that wouldn't be approved in wealthy countries.
Who are Christine Stabell Benn and Peter Aaby, and why are they controversial?
Benn and Aaby are Danish researchers who have published extensively on vaccine immunology and published contentious theories about alleged unintended vaccine effects. Their work suggests that some vaccines might have negative "non-specific effects" beyond their intended targets. However, their findings have not been replicated by independent research teams, and their methodology has been criticized by other scientists. Despite these concerns, Robert F. Kennedy Jr. has cited their work when questioning vaccine safety. This connection raised concerns about whether the CDC's funding of their trial was based on scientific merit or ideological alignment with Kennedy's skepticism about vaccines.
What is Robert F. Kennedy Jr.'s role in this controversy?
Robert F. Kennedy Jr. became Health Secretary in 2025 after a career promoting vaccine skepticism through his organization Children's Health Defense. He has consistently questioned vaccine safety based on theories contradicted by scientific evidence. Shortly after Kennedy took office, the CDC's advisory committee removed the universal hepatitis B birth dose recommendation, and funding flowed to researchers whose work Kennedy has publicly cited. These policy changes suggest Kennedy's ideology about vaccines influenced federal health decisions, creating apparent conflicts of interest in research funding.
Why is withholding a proven vaccine unethical in research?
Withholding a proven intervention without compelling justification violates international research ethics standards like the Declaration of Helsinki. These standards require that research benefit participants and don't deviate from proven methods without scientific uncertainty justifying the deviation. In this case, hepatitis B birth dose vaccination is proven effective, with no credible evidence of harm. Therefore, withholding it for research provides no compensating benefit to participants while exposing them to serious harm. This represents a straightforward ethical violation that international standards were designed to prevent.
How many countries use hepatitis B birth dose vaccination?
More than 115 countries have implemented hepatitis B birth dose vaccination as part of their national immunization schedules. This includes the United States, all European nations, most Asian countries, and many African nations. The global adoption reflects scientific consensus about the intervention's safety and effectiveness. The only reason Guinea-Bissau hadn't yet implemented it was resource constraints, not scientific doubt about its benefits. The government had already decided to implement it by 2028.
What is the current status of the trial?
As of early 2026, the trial was suspended in Guinea-Bissau pending ethical and technical review. The country's government decided insufficient coordination had occurred to approve the study. However, the US Department of Health and Human Services claimed the trial was "proceeding as planned," creating confusion about its actual status. The Africa Centres for Disease Control and Prevention suggested the trial would not proceed. Without Guinea-Bissau's participation, the trial cannot actually be conducted despite US claims that it continues.
Why does this matter beyond hepatitis B vaccine policy?
This situation represents a test of whether international research ethics standards will be upheld when challenged by powerful actors. Allowing this trial would establish a precedent that withholding proven interventions is acceptable in research if conducted in low-income countries. It would suggest that federal funding decisions can be influenced by ideology rather than scientific merit. It would undermine protections for vulnerable populations in international research. The outcome will influence how future ethically questionable research proposals are evaluated globally.
What would happen if the trial proceeded and confirmed birth dose safety?
If the trial proceeded and confirmed that hepatitis B birth dose vaccination is safe and effective, it would simply duplicate research that's already been conducted in numerous countries over three decades. The trial wouldn't provide new information because the safety question is already answered. This highlights that the trial wasn't primarily motivated by genuine scientific uncertainty. Instead, it appeared designed to generate data that could be used to question birth dose vaccination, regardless of actual findings.
How can countries protect their populations from ethically problematic research?
Host countries can protect their populations by maintaining strong ethics oversight systems that genuinely scrutinize foreign research. They should demand that international research meets the same standards it would need to meet in the researcher's home country. They should engage with international oversight organizations like WHO when ethical concerns arise. They should be willing to refuse permission for studies that don't serve their population's interests, regardless of financial incentives. Guinea-Bissau's suspension of this trial demonstrates how this protection works in practice.
What role should international organizations like WHO play in research oversight?
The WHO's formal statement about this trial demonstrates the appropriate role for international organizations. When national research ethics systems fail or when powerful interests override protections for vulnerable populations, international oversight becomes critical. The WHO can articulate scientific and ethical standards that transcend national politics. However, international oversight works best when combined with strong national oversight. Host countries must retain ultimate authority over research conducted on their populations, while international organizations provide standards and expertise.

The Path Forward: Ensuring Research Integrity in Vaccine Science
The hepatitis B vaccine trial controversy will likely have lasting effects on how vaccine research is conducted and funded. The public attention it received, the international condemnation it faced, and the host country's willingness to suspend it all demonstrate that violations of research ethics standards will be challenged.
For the scientific community, this situation reaffirms the importance of research integrity. Funding decisions must be based on scientific merit, not ideology. Researchers must design studies that pass ethical review genuinely, not just formally. International oversight must be strengthened to prevent vulnerable populations from being used for ethically questionable research.
For policymakers, the lesson is that vaccine policy decisions must remain grounded in evidence. Decades of global experience with hepatitis B vaccination provide clear evidence about benefits and safety. New policies should build on this evidence, not contradict it based on controversial theories from fringe researchers. Appointing officials skeptical of vaccines to lead health agencies creates predictable conflicts of interest in research funding and policy decisions.
For global health organizations, the challenge is maintaining commitment to evidence-based recommendations despite pressure from powerful countries pursuing different policies. The WHO's condemnation of this trial showed that international health bodies can stand firm. However, they need support from other countries, from medical professionals, and from engaged citizens who demand that public health be based on evidence.
The hepatitis B vaccine birth dose will almost certainly become standard practice in Guinea-Bissau by 2028, as the government already decided. Thousands of Guinean infants will be protected from mother-to-child hepatitis B transmission. The birth dose will prevent chronic infection, liver disease, and cancer in their lives. This represents the outcome that evidence supports and ethics demands.
The trial's suspension, whether permanent or temporary, represents a victory for established research ethics standards and international health principles. It demonstrates that concerned scientists, public health organizations, and governments can collectively prevent ethically problematic research from proceeding, even when it's funded and advocated by powerful actors. That precedent matters for protecting vulnerable populations globally and ensuring that vaccine science remains focused on evidence rather than ideology.
As the world's vaccine policies continue to evolve, the principles demonstrated by this controversy must remain central. Research ethics aren't obstacles to scientific progress—they're requirements for conducting research responsibly. Withholding proven interventions without justification doesn't advance science. It violates the foundational ethical commitments that make medical research trustworthy. Maintaining those standards protects both individual research participants and public trust in vaccine science globally.

Key Takeaways
- The WHO formally condemned a US-funded trial for withholding hepatitis B vaccine birth doses from 14,000 newborns in Guinea-Bissau, violating international research ethics standards
- Hepatitis B birth dose vaccination prevents mother-to-child transmission in 95%+ of cases and is used by 115+ countries with three decades of proven safety data
- The CDC awarded $1.6M to controversial researchers Christine Stabell Benn and Peter Aaby despite their findings not being replicated and Robert F. Kennedy Jr.'s known skepticism about vaccines
- The trial's design violated multiple ethical principles: withholding proven intervention, single-blind methodology, lack of active control group, and exploiting resource constraints in a poor country
- Guinea-Bissau suspended the trial pending ethical review, demonstrating host countries can protect their populations from ethically problematic research despite funding incentives
![WHO Condemns US Vaccine Trial Ethics: The Guinea-Bissau Hepatitis B Controversy [2025]](https://tryrunable.com/blog/who-condemns-us-vaccine-trial-ethics-the-guinea-bissau-hepat/image-1-1771025768212.jpg)



