Introduction: When AI Meets Drug Discovery
Imagine spending 10 to 15 years and over $2.6 billion to bring a single drug to market, only to have it fail in clinical trials. That's the current reality of traditional pharmaceutical drug discovery. Researchers screen thousands of molecular compounds through a process called high-throughput screening, which is expensive, time-consuming, and surprisingly ineffective. The failure rate is staggering. For every 5,000 to 10,000 compounds screened, typically only one makes it through the entire development pipeline to FDA approval.
But what if artificial intelligence could change that equation entirely?
Enter Chai Discovery, a startup founded in 2024 that's already become one of the most talked-about names in the AI-powered biotech space. In just over a year, the company has raised hundreds of millions of dollars, attracted backing from some of Silicon Valley's most influential venture capitalists, and most importantly, secured a landmark partnership with Eli Lilly, one of the world's largest pharmaceutical companies. This isn't hype—it's a fundamental shift in how molecules are designed.
What makes Chai's story fascinating isn't just the money or the partnerships. It's how the company emerged from conversations between its founders and OpenAI's Sam Altman, how the founders spent years waiting for the technology to mature, and how they finally seized the moment when the conditions were right. The startup represents something bigger: a convergence of AI capability, computational power, and biological understanding that's reaching a critical inflection point.
This article explores how Chai Discovery went from concept to unicorn, what their technology actually does, why pharmaceutical giants are betting billions on AI-driven drug discovery, and what this means for the future of medicine. We'll dive deep into the team, the technology, the partnerships, the skepticism, and the enormous potential.
TL; DR
- **Chai Discovery raised 1.3 billion valuation in just 14 months of operation as reported by The Healthcare Technology Report.
- Eli Lilly partnership announced to use Chai's Chai-2 algorithm for antibody development, with expectations for first-in-class medicines in clinical trials by 2027 according to Yahoo Finance.
- Founders came from OpenAI, Facebook, and Absci, bringing expertise in AI models, protein folding, and biotech drug discovery as detailed by TechCrunch.
- AI-driven drug discovery could compress timelines from 10-15 years to potentially 5-7 years, according to industry projections as noted by IBM.
- The market opportunity is massive: companies that adopt these technologies early could achieve significant competitive advantages and bring life-saving medicines to patients faster according to McKinsey.

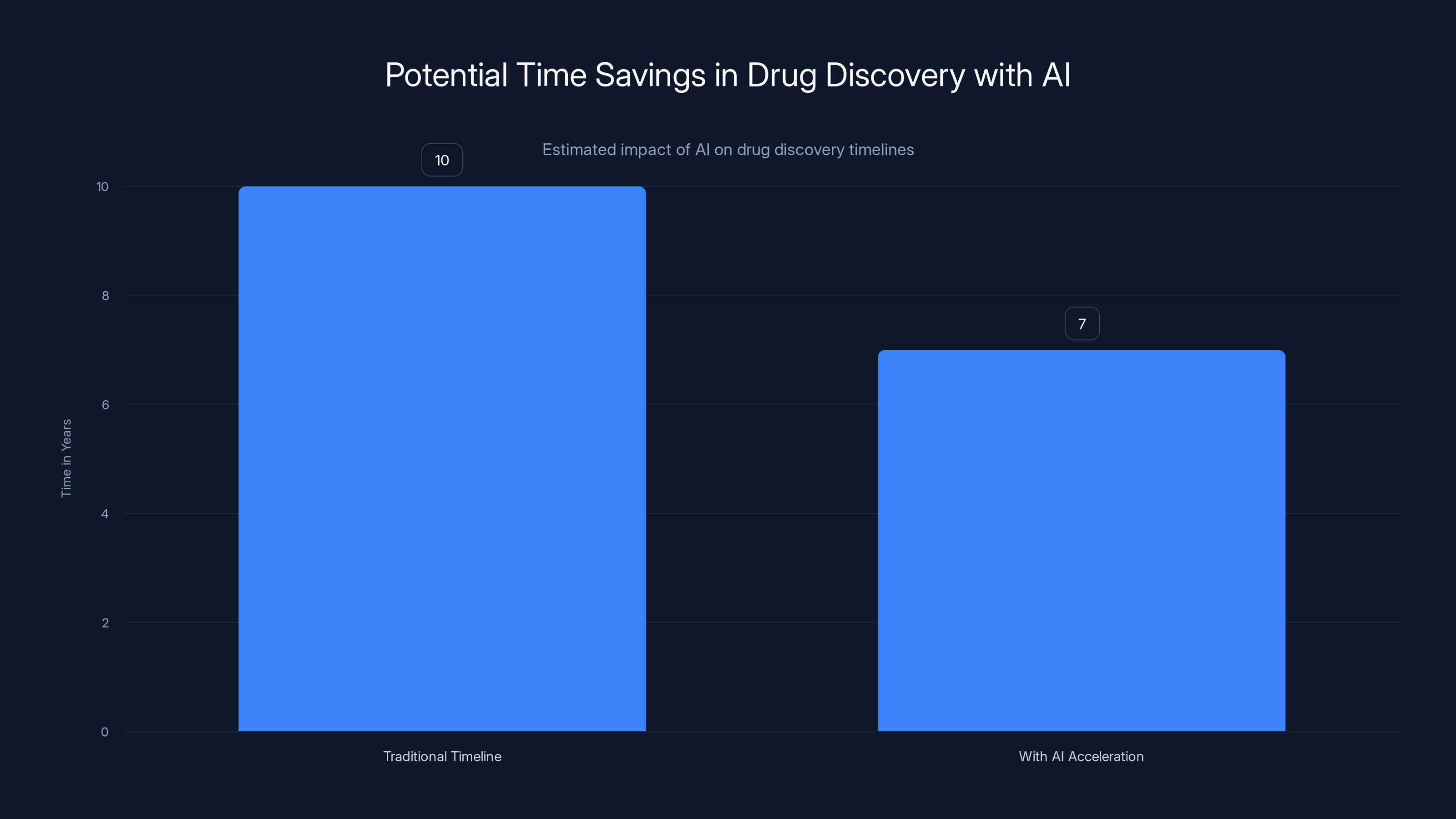
AI tools can potentially reduce the drug discovery timeline by 1-3 years, accelerating the process from 10 years to approximately 7 years. Estimated data.
The Founders: How Conversations With Sam Altman Sparked a Biotech Revolution
The origin story of Chai Discovery reads like a Silicon Valley screenplay. It begins not in 2024, when the company was officially founded, but six years earlier, when conversations started between Sam Altman, CEO of OpenAI, and Josh Meier, who was working on OpenAI's research and engineering team in 2018.
Meier eventually left OpenAI, and Altman reached out to Meier's college friend from Harvard, Jack Dent, who at the time was working as an engineer at Stripe. Altman had a specific proposition: would they be interested in starting a proteomics company? Proteomics is the study of proteins, and it's central to drug discovery. Proteins are the workhorses of biology—they do the actual work in our bodies, and understanding them is fundamental to creating new medicines.
Dent was intrigued, but Meier wasn't ready. The AI technology needed for such a venture simply wasn't advanced enough. As Dent recalls, "Josh and I thought the technology wasn't there yet. It was still too early." This wasn't hesitation born from fear—it was pragmatism. Meier could have started the company in 2018 with much less competition, but he knew the odds of success would be lower. Instead, he joined Facebook's research team.
At Facebook (now Meta), Meier worked on something that would become crucial to Chai's future: ESM1, the first transformer-based protein language model. This was groundbreaking. Just as language models like GPT can predict the next word in a sentence by understanding patterns in billions of words, protein language models can predict protein structures and properties by understanding patterns in millions of protein sequences. This technology became a direct precursor to what Chai would later build.
After Facebook, Meier spent three years at Absci, another AI biotech company focused on drug discovery. This gave him deep hands-on experience in how AI could actually be applied to real drug development problems. He learned what works, what doesn't, and where the biggest opportunities lie.
By 2024, both Meier and Dent felt the time was right. The AI technology had matured. The computational infrastructure was in place. The scientific understanding was deeper. They reached back out to Sam Altman and said, "Let's do this." Altman agreed to become one of Chai's seed investors, and more importantly, he let the founders work out of OpenAI's San Francisco offices in the Mission District while they were getting started.
Two other co-founders joined the team: Matthew Mc Partlon and Jacques Boitreaud, bringing additional expertise in computational biology and machine learning. The team was small but exceptionally well-credentialed. These weren't first-time founders trying to guess their way through biotech. They were scientists and engineers who had spent years at the companies that built modern AI and understood the frontier of what was possible.
The Chai-2 Algorithm: Designing Molecules Like Software
At the core of Chai Discovery's value proposition is a piece of software called Chai-2. This isn't a minor tool—it's the intellectual property that attracted Eli Lilly's attention and justified a billion-dollar valuation.
Chai-2 is designed to do something that sounds simple but is extraordinarily complex: generate antibodies. Antibodies are proteins that our immune system produces to fight disease. They're also increasingly important as medicines. Many of the most successful modern drugs are actually antibodies that have been engineered to target specific diseases. Cancer medications, autoimmune treatments, and biological therapies rely heavily on antibody design.
Traditionally, finding the right antibody for a given disease requires screening libraries of millions or billions of candidates. Scientists test them one by one or in batches, looking for the ones that bind to the target protein effectively. It's slow. It's expensive. It's hit-or-miss.
Chai-2 approaches this differently. It uses generative AI—the same kind of technology that powers large language models—to design antibodies from scratch. You give the algorithm a target protein (the thing you want to attack), and it generates candidate antibodies designed to bind to that target with high specificity. The algorithm has learned patterns from vast amounts of protein sequence and structure data, and it uses those patterns to predict what sequences will work best.
But here's the crucial part: Chai-2 doesn't just generate random sequences. It generates molecules that are designed to be manufacturable, stable, and effective. It understands not just the biology but the practical constraints of pharmaceutical production. This is what distinguishes it from a generic AI tool applied to biology.
The algorithm works somewhat like this: imagine you're designing a lock, and you need to create the perfect key to fit it. A brute-force approach would be to try billions of keys. Chai-2 is more like a designer who understands key-making so well that it can generate keys that have a very high probability of fitting on the first try. This compression of the design space from millions of possibilities to a smaller set of high-quality candidates dramatically speeds up the discovery process.
Jack Dent described the company's aspiration as building a "computer-aided design suite" for molecules. Think of it like CAD software for molecules. Just as architects use CAD tools to design buildings and iterate rapidly without building physical prototypes, Chai is building tools that let drug designers iterate rapidly on molecular designs without synthesizing hundreds of physical compounds.
The mathematics underlying this kind of generative design is fascinating. When you're designing antibodies, you're working with a sequence of amino acids, typically around 110-130 amino acids long, depending on the region. Each position can be one of 20 different amino acids. That's 20^110 possible combinations—a number so large it exceeds the number of atoms in the observable universe. You can't search this space exhaustively. You need intelligence to navigate it. Generative models trained on natural antibodies learn the high-probability regions of this sequence space, the regions where functional antibodies are likely to exist. By sampling from these learned distributions, the algorithm can generate candidate antibodies that are much more likely to work than random sequences.

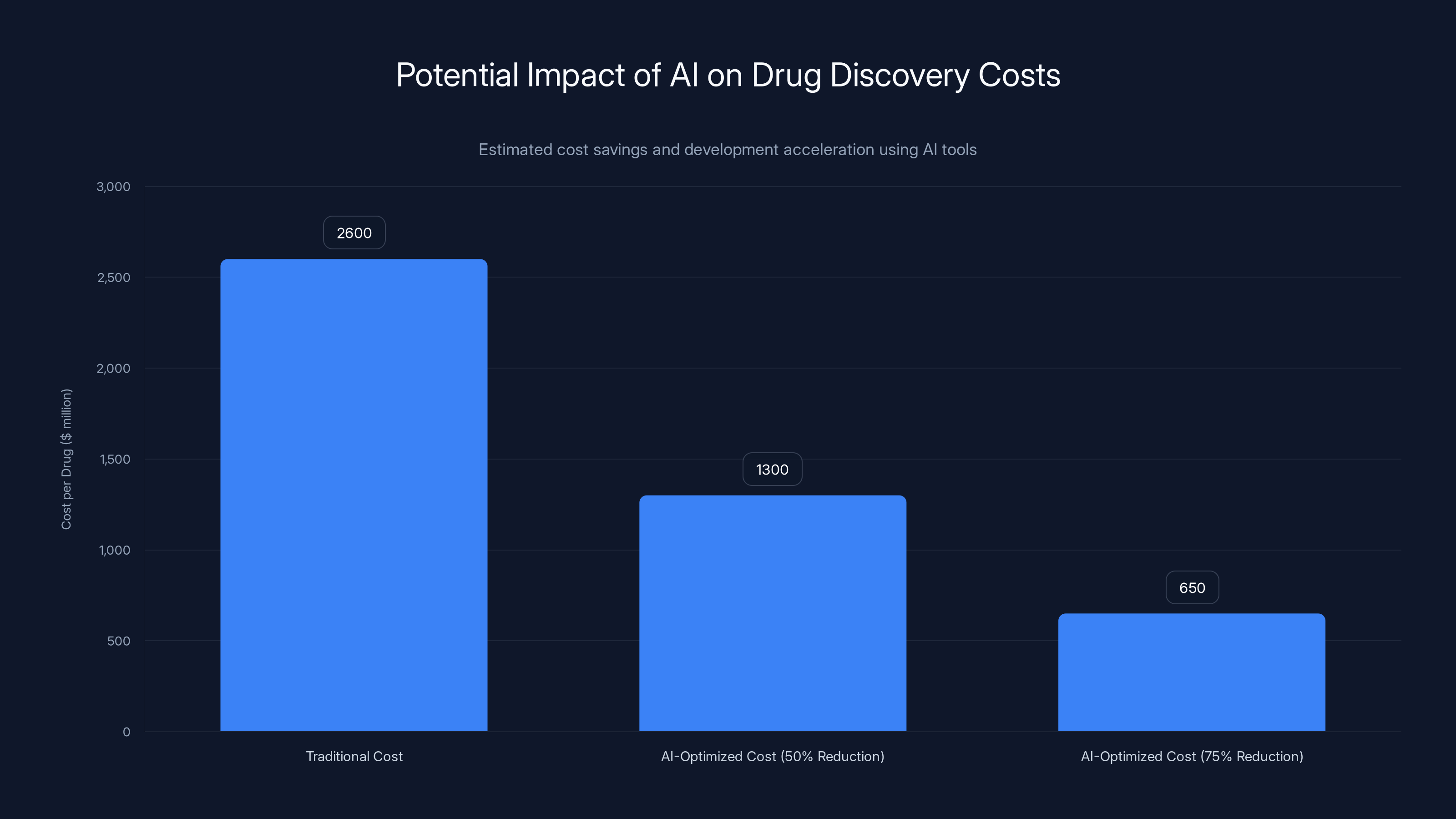
AI tools could significantly reduce drug development costs, potentially cutting them by 50% to 75%. Estimated data.
The Valuation Sprint: From Seed to Unicorn in 14 Months
Chai Discovery's funding trajectory has been nothing short of remarkable. The company was founded in 2024 and closed its Series B round in December 2024, just over a year later, having raised a combined total in the hundreds of millions of dollars with a valuation of $1.3 billion.
Let's put that in perspective. Most companies take three to five years to reach a $1 billion valuation. Chai did it in 14 months. This isn't unprecedented—some AI companies in hot markets have moved similarly fast—but it's a strong signal that investors see massive potential in the space.
The Series B round brought in $130 million in fresh capital. Major investors include General Catalyst, a top-tier venture firm with deep expertise in biotech and AI, along with other leading venture capitalists from Silicon Valley. OpenAI, which had been an early seed investor, continued to support the company.
What's notable is that these investors aren't betting on hype. General Catalyst's Elena Viboch explicitly stated the firm's thesis: companies that move quickly to partner with AI-driven drug discovery platforms like Chai will be the first to get molecules into clinical trials. She expressed confidence that partnerships formed in 2026 could result in first-in-class medicines entering clinical trials by the end of 2027 as noted by AlleyWatch.
That's a specific, measurable claim. Viboch isn't saying "maybe this will work someday." She's saying "we expect to see clinical results in 18-24 months." For a venture capitalist to make that claim publicly is significant. It suggests the firm has done deep diligence and believes the technology is at an inflection point.
The speed of this funding tells us something important about market timing. Proteomics startups have been attempted before. Why is Chai getting billion-dollar valuations in year one, while previous attempts took longer to gain traction? The answer is that the underlying AI technology—transformer models, large language models, generative AI—has become powerful enough and cheap enough to actually transform drug discovery. The computational infrastructure is in place. The training data is available. The tools are mature. It's not that the idea is new; it's that the enabling technology has finally caught up to the vision.
The Eli Lilly Partnership: Validation From the Pharmaceutical Establishment
On the same day Chai announced its Series B funding, it also announced something arguably more valuable: a partnership with Eli Lilly, one of the world's largest and most respected pharmaceutical companies. This isn't a small validation—this is the pharmaceutical equivalent of a major tech company endorsing a developer platform.
Eli Lilly is a 148-year-old company with a market cap in the hundreds of billions. It's not a venture-backed startup taking wild bets. When Eli Lilly signs a strategic partnership, it means the technology has cleared serious internal scrutiny. The company has likely run simulations, consulted with its scientific teams, and determined that this partnership could genuinely accelerate their drug discovery process as reported by Yahoo Finance.
Under the partnership agreement, Eli Lilly will use Chai's generative design models in combination with Lilly's internal expertise in biologics (protein-based drugs) and its proprietary datasets. The goal is to design better molecules from the outset—to use AI to generate antibody candidates that are more likely to be successful, and to generate more of them, faster.
Aliza Apple, the head of Lilly's Tune Lab program (which focuses on using AI and machine learning for drug discovery), said the partnership aims to "push the frontier of how AI can design better molecules from the outset, with the ultimate goal to help accelerate the development of innovative medicines for patients."
This partnership is strategic in multiple ways. First, it provides Chai with real-world validation. When Chai's algorithms are used at Eli Lilly scale, the results will either validate the technology or expose limitations. That real-world data is invaluable for refining the algorithm. Second, it provides Eli Lilly with early access to a technology that could give it a competitive advantage in drug discovery. If Chai's tools genuinely compress discovery timelines, Lilly's pipeline will move faster than competitors using traditional methods. Third, it creates a proof point that's worth billions. If Chai's algorithms help Eli Lilly bring even one drug to market months or years faster, the ROI could be enormous.
Consider the economics. If a blockbuster drug generates $2 billion in annual revenue, and Chai's tools help bring that drug to market one year earlier, the present value of that accelerated cash flow could be hundreds of millions of dollars. Eli Lilly is a rational actor. It wouldn't sign this partnership if the potential upside wasn't substantial.
The partnership also highlights an important trend: pharmaceutical companies are moving from skepticism to partnership on AI. Five years ago, many traditional pharma executives viewed AI with skepticism. The technology was untested in drug discovery. Would it really work? Today, pharma companies are partnering with AI startups, not just funding research, but actually integrating AI tools into their discovery pipelines as noted by the Grand Forks Herald.

The Industry Landscape: Who Else Is Building AI Drug Discovery Tools?
Chai Discovery isn't the only player in AI drug discovery. There's a growing ecosystem of companies approaching the problem from different angles. Understanding the competitive landscape helps explain why Chai's positioning is unique.
Some companies focus on protein folding prediction. DeepMind's AlphaFold was a breakthrough—it could predict protein 3D structures from amino acid sequences with unprecedented accuracy. This is foundational research that makes other drug discovery work easier. But AlphaFold is a research tool, not a drug discovery platform. It predicts structures; it doesn't design molecules.
Other companies focus on specific disease areas or molecule types. Some specialize in small molecule drug design (traditional chemical drugs), while others, like Chai, specialize in biologics (protein-based drugs). The landscape is fragmented because drug discovery is incredibly complex. A tool that works brilliantly for antibodies might not work for small molecules. A tool that works for oncology might not work for autoimmune diseases.
What sets Chai apart is its combination of three things: first, the founders' deep experience in both AI (from OpenAI and Facebook) and biotech (from Absci). Second, the focus on antibodies and biologics, which is one of the hottest areas in pharmaceutical development. Third, the early partnerships and validation from major pharma players.
The competitive advantage isn't just technical; it's also network-based. When Eli Lilly partners with Chai, it gives Chai access to Lilly's data, expertise, and clinical network. That's a moat. A competitor would have to build similar partnerships with other pharma companies, and those deals take time and proof of concept.
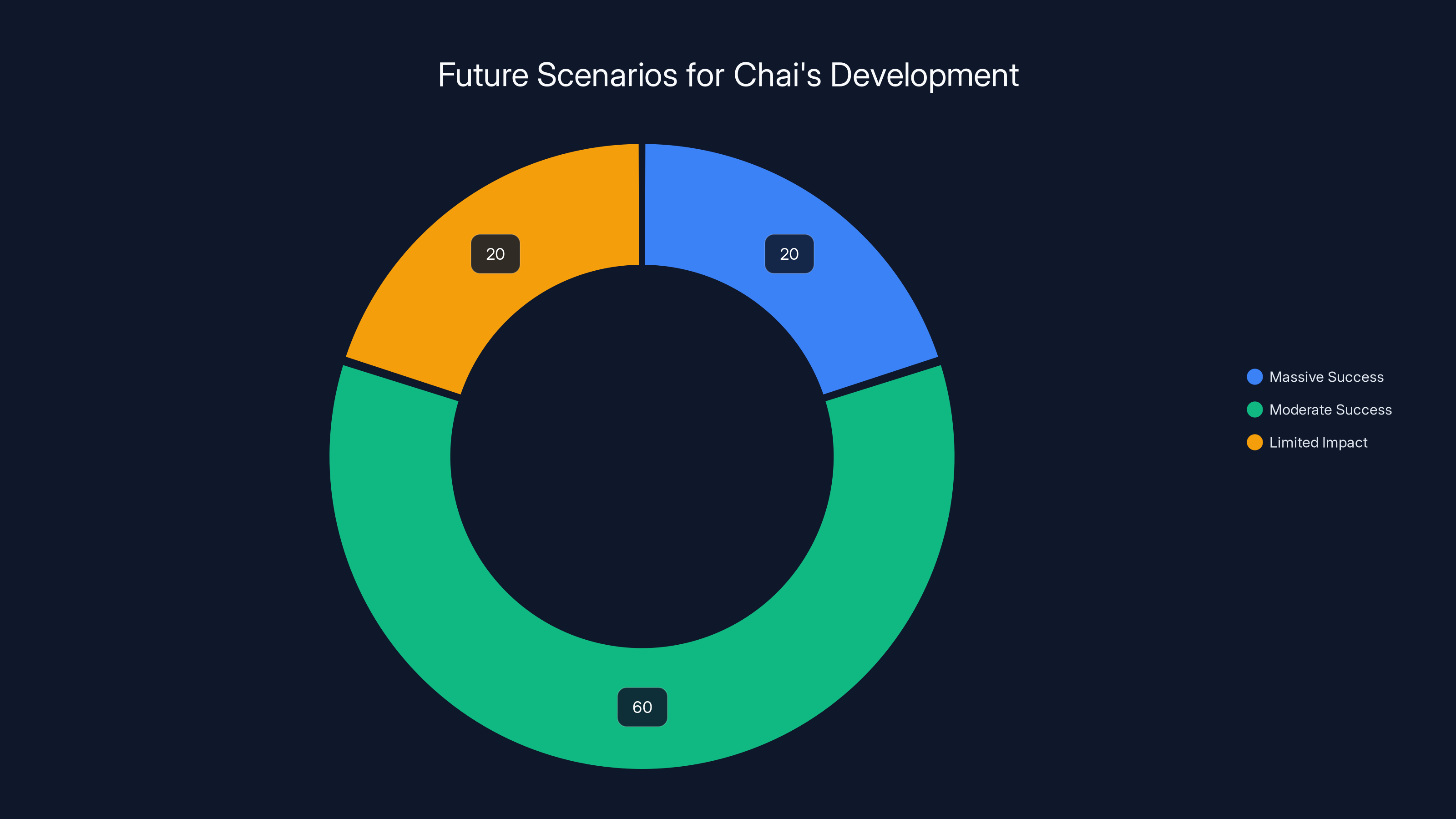
Estimated data suggests that 'Moderate Success' is the most likely outcome for Chai, with a 60% chance, while 'Massive Success' and 'Limited Impact' both have a 20% likelihood.
The Technology Evolution: From Theoretical AI to Practical Drug Discovery
To understand why now is the right moment for Chai, we need to zoom out and look at the evolution of AI capabilities over the past decade.
In 2015, deep learning breakthroughs were happening in image recognition and natural language processing. The ImageNet competition had been won by deep neural networks. But biology was still largely unaffected by AI. Pharma companies were still using traditional methods: high-throughput screening, medicinal chemistry, structural biology.
In 2017, transformer models were introduced. This architecture became the foundation for all modern large language models. Transformers are particularly good at learning patterns in sequences—which is exactly what proteins are: sequences of amino acids. By 2020, researchers started applying transformer models to protein sequences, and the results were startling. The models could learn useful representations of proteins from unlabeled sequence data.
In 2020, AlphaFold shocked the scientific community by solving the protein structure prediction problem. This demonstrated that deep learning, when applied to biology, could achieve breakthroughs that traditional structural biology hadn't achieved in 50 years.
Simultaneously, the availability of training data improved. Public protein sequence databases grew larger. Computational resources became cheaper. GPUs and TPUs that were expensive and rare became commodities available via cloud providers.
By 2023-2024, the pieces were in place. Transformer models were mature. Protein language models had been developed and refined. Large foundation models trained on broad data were showing unexpected capabilities. The computational infrastructure was ready. The data was available. The talent pool of people who understood both deep learning and biology was growing.
This is why Josh Meier was right to wait. In 2018, the technology simply wasn't mature enough. In 2024, everything had converged. The founders could build on years of previous research—AlphaFold, protein language models, generative models—rather than inventing fundamentals as discussed in Nature.

The Skeptics: What Could Go Wrong?
Not everyone is convinced that AI will revolutionize drug discovery. Some industry veterans remain skeptical, and it's worth understanding why.
One concern is that drug discovery is fundamentally harder than people realize. Generating a candidate molecule is one thing. Getting that molecule through clinical trials and FDA approval is another. Many molecules that look great computationally fail in practice. They might be toxic, or they might not work as expected in living organisms. Proteins don't exist in isolation; they work in complex biological systems.
Another concern is that the hype cycle around AI has a track record of overshooting reality. Every few years, there's a new AI technology that's supposed to transform medicine. Some of these claims have panned out; others have not. Skeptics worry that generative AI for drug discovery is the next overhyped frontier.
A third concern is data quality. Machine learning models are only as good as the data they're trained on. If the training data has biases or errors, the model will amplify those biases. Drug discovery data is expensive and proprietary. Training a model on limited or biased data could lead to bad predictions.
These are all legitimate concerns. They deserve to be taken seriously. But it's also worth noting that skepticism can be the enemy of progress. AI protein design tools have already made contributions to drug discovery. Generative models have generated candidate molecules that were synthesized and tested in the lab, and some have shown promise. The skeptics might be right that we won't see a wholesale transformation overnight, but the early signs suggest real progress.
The beauty of Chai's partnership with Eli Lilly is that these concerns will be empirically tested. If Chai's tools help Eli Lilly generate better molecules and accelerate development, the results will speak for themselves. If the tools overpromise and underdeliver, that will become apparent too. The market will provide feedback.
The Broader Market: How Big Could This Be?
To understand Chai's potential, consider the size of the drug discovery market and the potential for cost savings and acceleration.
On average, bringing a new drug to market costs $2.6 billion and takes 10 to 15 years. This includes all the failures—the compounds that didn't work, the clinical trials that failed, the regulatory setbacks. For every drug that makes it to market, thousands don't.
The discovery phase—the process of identifying a drug candidate and proving it works in the lab—typically takes 3 to 6 years. If AI tools could compress this to 6 months to 1 year, the time savings would be substantial. For a blockbuster drug generating $2 billion in annual sales, moving the launch date forward by one year could be worth hundreds of millions of dollars in present value.
There are roughly 7,000 to 10,000 new molecular entities in development globally at any given time. If even 10% of those could be developed faster or more cost-effectively using AI tools, that's 700 to 1,000 drugs. At an average cost of
Furthermore, the impact isn't just on cost savings. It's on the types of drugs that become economically viable. Some diseases are hard to treat because no good drug candidates have been found, not because they're fundamentally undruggable, but because the search space is too large to explore with traditional methods. AI tools that can navigate this space might unlock treatments for diseases that have been intractable.
Consider rare diseases. There are thousands of rare genetic diseases affecting millions of people worldwide. Each disease has a small patient population, so the market isn't big enough to justify traditional drug development economics. But if AI tools could compress discovery timelines and reduce costs by 50% or 75%, suddenly some of these rare disease programs become economically viable. That's not just a business opportunity; it's a humanitarian opportunity.

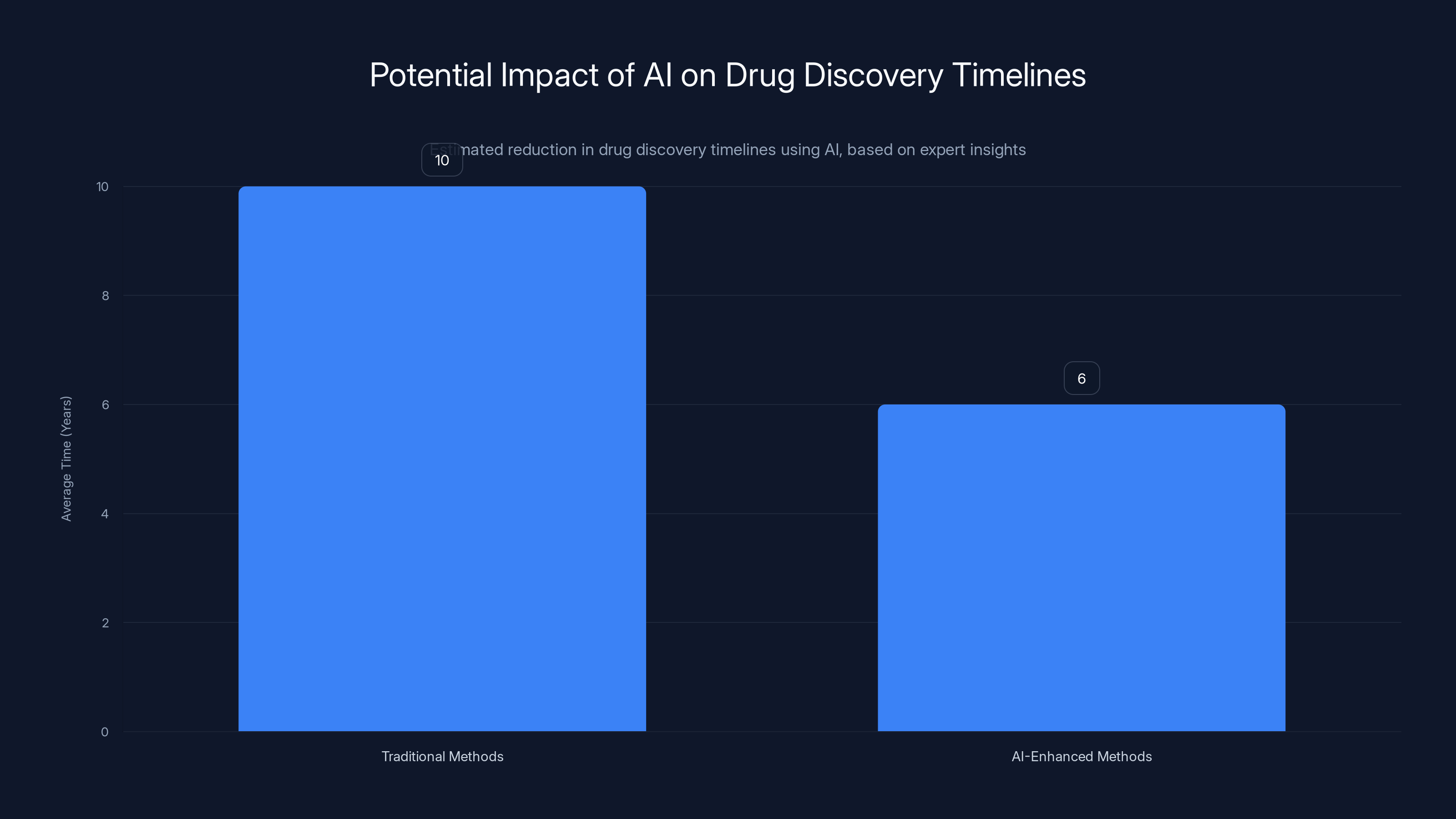
AI-enhanced methods, like those used by Eli Lilly and Chai, could potentially reduce drug discovery timelines by up to 40%, accelerating the development of innovative medicines. Estimated data.
Capital Formation and Investor Thesis
Why are venture capitalists throwing hundreds of millions of dollars at Chai Discovery and similar companies? Understanding the investor thesis helps explain the valuations.
General Catalyst's investment thesis, as articulated by Elena Viboch, is that "biopharma companies that move the most quickly to partner with companies like Chai will be the first to get molecules into the clinic, and will make medicines that matter." This is a clear competitive advantage narrative. The investors believe that using AI-powered drug discovery tools creates a first-mover advantage in pharma. The company that gets a molecule into clinical trials first has a window of opportunity—it can start generating clinical data, demonstrating efficacy, building a regulatory narrative. Competitors using traditional discovery methods will be years behind.
This thesis has precedent. In genomics, companies that adopted next-generation sequencing early gained competitive advantages. The sequencing landscape shifted from Sanger sequencing (slow and expensive) to NGS (fast and cheap) within a decade. Companies and institutions that embraced NGS early were able to do research that was economically impossible with older technology. The same dynamic could play out in drug discovery.
Another part of the investor thesis is the network effect. As Chai partners with more pharmaceutical companies, it gets more data, more feedback, and more validation. The algorithm improves. The tool becomes more valuable. This creates a moat around Chai—competitors would struggle to catch up because they don't have access to the data flowing from multiple pharma partnerships.
There's also the portfolio argument. For a VC firm investing in healthcare, having a successful position in an AI drug discovery platform is attractive. The upside could be enormous. If Chai's tools become standard in pharma, the company could be acquired by a larger biotech or pharmaceutical company at a substantial premium, or it could go public at a high valuation. The downside is that the company doesn't work out and the investors lose their investment. But the upside/downside ratio looks attractive to investors who believe in the AI-biotech convergence.
It's also worth noting that late-stage venture funding in AI is abundant. There's a lot of capital looking for places to invest, and biotech is seen as a high-impact sector. The conjunction of easy capital, AI breakthroughs, and pharma interest creates conditions for rapid capital formation in this space.
Technical Depth: How Generative Models Work for Molecular Design
For readers interested in the technical details, let's dig deeper into how generative models actually design molecules.
The core concept is that you can represent a protein or antibody as a sequence of tokens (amino acids), just like you can represent text as a sequence of tokens (letters and words). A transformer model trained on many protein sequences learns to predict the next amino acid in a sequence, given the previous amino acids. This is the same principle as language models predicting the next word.
But predicting the next token in a random sequence isn't useful. What you want is to generate sequences that have desirable properties: they bind to a target, they're stable, they're manufacturable. This requires a more sophisticated approach.
One approach is diffusion models. A diffusion model works backwards from the goal. You start with a random sequence and iteratively denoise it, guided by a model that understands what good antibodies look like. At each step, the model adds information that makes the sequence more likely to be a good antibody. After enough iterations, you have a high-quality candidate.
Another approach is latent space modeling. You train a model that compresses proteins into a lower-dimensional representation (a latent space). In this latent space, you can navigate smoothly: small changes in the latent space correspond to small changes in protein properties. You can then use optimization techniques to find regions of latent space where antibodies have the properties you want, and decode those latent representations back into protein sequences.
A third approach is reinforcement learning. You train a generative model to produce sequences, and you reward it for sequences that have desirable properties (binding affinity, stability, manufacturability). Over time, the model learns to generate better and better sequences.
In practice, successful drug discovery AI likely combines multiple approaches. A generative model creates candidates. A scoring function evaluates them based on predicted properties. A selection mechanism chooses the most promising candidates for synthesis and testing. The feedback from real-world testing informs the next iteration of the model.
The mathematical formulation involves optimizing over the sequence space to maximize a multi-objective function:
where
The key insight is that all of this happens in silico, in the computer, before any molecules are actually synthesized. This is what makes it so fast. Instead of synthesizing 10,000 candidates and testing each one, you generate 10,000 candidates computationally, score them, select the top 100, synthesize those, and test those. You've compressed the search space using intelligence.

The Timeline Question: Can AI Really Accelerate Drug Discovery?
One of the boldest claims made by Chai's investors is that partnerships formed in 2026 could result in first-in-class medicines in clinical trials by 2027. That's an 18-24 month timeline from partnership to clinical trials.
Is this realistic? Let's break down a typical drug discovery timeline:
-
Target identification and validation (6-24 months): Identify the biological target you want to hit. Prove that hitting this target will actually treat the disease. This phase typically can't be accelerated much by computational tools.
-
Hit identification (3-6 months): Find initial compounds that bind to the target, even weakly. This is where AI tools can help. Instead of screening millions of compounds experimentally, you can generate and computationally screen thousands of candidates, then synthesize the top ones for experimental validation.
-
Lead optimization (2-4 years): Take a promising hit and optimize it: improve binding affinity, reduce off-target effects, improve pharmacokinetics (how the body processes it), improve manufacturability. This is a long phase and often the bottleneck. But AI tools can accelerate this too by suggesting chemical modifications that improve specific properties.
-
Preclinical testing (3-6 years): Test in cell cultures and animal models. Demonstrate safety and efficacy before human trials.
-
IND application and clinical trials (2-10 years): Human testing in phases I, II, and III. This phase is hard to accelerate because it depends on recruiting patients, running trials, and collecting safety and efficacy data.
The claim that Eli Lilly could have a first-in-class medicine in clinical trials by 2027 likely assumes that Eli Lilly is starting with a disease target they've already validated. They already know what to attack. Chai's tools would accelerate hit identification and lead optimization—potentially compressing 2-4 years into 6-12 months. Then preclinical testing (which can happen in parallel with lead optimization) takes another 1-2 years. So 18-24 months to IND application is plausible if everything goes well.
But "if everything goes well" is a big caveat. Discovery projects fail for many reasons. The target might be harder to hit than expected. Candidates might have toxicity issues. Preclinical data might not look good. The timeline could easily stretch to 3-5 years.
The realistic expectation is probably: AI tools can compress drug discovery timelines by 20-50%, depending on the disease area and how well the AI tools work for that specific problem. This is significant—it's worth billions of dollars to pharma companies—but it's not a panacea. Drug discovery will still be a slow, expensive, uncertain business. But it will be less slow, less expensive, and somewhat less uncertain.
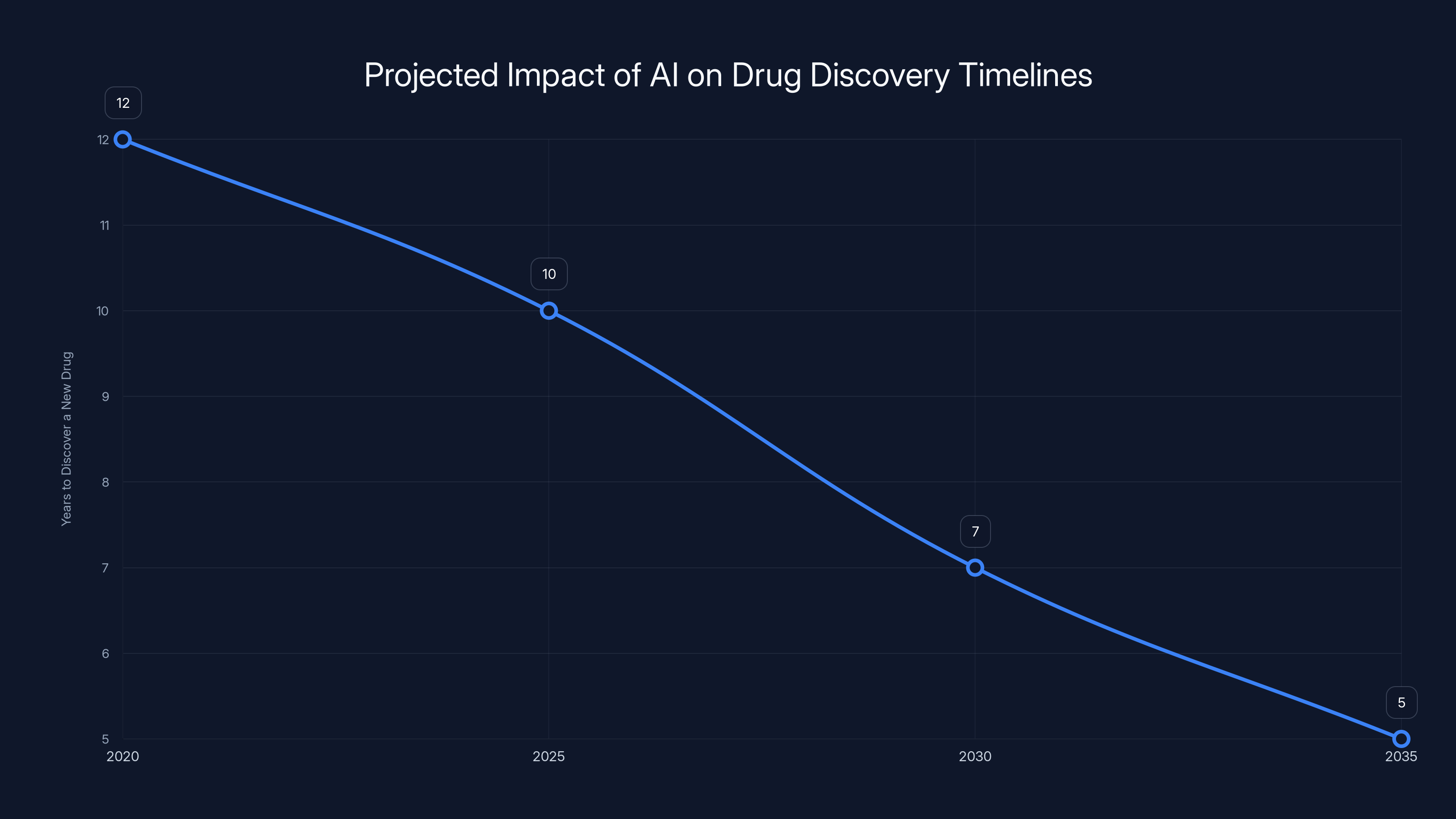
The integration of AI in drug discovery is projected to significantly reduce the time required to discover new drugs, potentially cutting timelines from 12 years in 2020 to just 5 years by 2035. (Estimated data)
Data and Proprietary Advantages
One of the most valuable assets in AI-driven drug discovery is proprietary data. The companies with the most data—about which drug candidates worked, which failed, and why—will have the best models.
Pharmaceutical companies sit on vast libraries of proprietary data about drug molecules: which ones bound to targets, which ones were toxic, which ones made it through clinical trials, and which ones failed. This data is a gold mine for training better AI models. It's also fiercely guarded. Most pharma companies won't share this data broadly.
This creates an interesting dynamic for Chai. The company doesn't own a library of proprietary molecules the way Eli Lilly does. But through partnerships, Chai gets access to anonymized, aggregated learnings from many drug discovery projects. This gives Chai's models exposure to more diverse types of problems than any single pharma company sees. A model trained on data from antibody programs, small molecule programs, and biologics programs will be more robust than a model trained only on antibodies.
Conversely, Eli Lilly gets to use Chai's general models trained on diverse data, and then specialize those models using Lilly's proprietary data. This partnership structure aligns incentives: both companies benefit from better models, and both companies protect their most sensitive IP.
Other companies trying to compete with Chai face a data disadvantage. They don't have Eli Lilly's proprietary data. Building a competitive model requires either: (1) training on public data only, which is limited, or (2) signing partnerships with other pharma companies to get access to their data. Chai has a head start with Eli Lilly. Competitors will be working to catch up.
The International Landscape and Regulatory Considerations
Chemistry and drug discovery aren't bound by national borders. Chai could potentially serve customers globally. But regulatory considerations, IP laws, and competition from regional players will shape the landscape.
In Europe, there are biotech companies and AI startups working on similar problems. The EU has been investing heavily in AI research and biotech. Some of these companies might compete with Chai or partner with it.
In China, there's growing interest in AI-driven drug discovery. Chinese biotech companies and AI labs are working on similar problems. The question is whether Chinese companies will develop equivalent technology, or whether the intellectual property advantages and partnerships that Western companies like Chai have built will be durable.
Regulatory approval of drugs is a global process. A drug approved by the FDA in the US, the EMA in Europe, and the PMDA in Japan will have a much larger addressable market than a drug approved in only one region. Companies like Chai that can serve global pharma customers have an advantage. But this also means regulatory standards matter. If AI-designed molecules face skepticism from regulators, it could slow adoption.
So far, there's no evidence of major regulatory pushback against AI-designed drugs. The FDA has been supportive of technologies that could accelerate drug development as reported by MedShadow. But this is early. As AI-designed drugs move into later-stage trials and seeking approval, we'll see how regulators handle novel molecules designed entirely by AI.
Future Scenarios: Three Possible Outcomes
Looking forward, there are several plausible scenarios for how Chai's story unfolds:
Scenario 1: Massive Success (Most Optimistic)
Chai's tools prove transformative. Within 5 years, major pharma companies are using Chai's platform for 20-30% of their new drug discovery projects. The company becomes the dominant provider of AI drug design tools in the biologics space. Chai goes public at a $10-20 billion valuation or is acquired by a large pharma company for a substantial premium. Multiple blockbuster drugs are brought to market faster because of Chai's tools. The company becomes a household name in biotech.
Scenario 2: Moderate Success (Most Likely)
Chai's tools prove useful but not revolutionary. They accelerate drug discovery by 20-40%, providing real value to partner companies. The company remains valued at $2-5 billion. It goes public or is acquired at a solid return for investors. Chai is one of several successful players in the AI drug discovery space, competing with other startups and established players. Innovation continues incrementally, with each new tool providing modest improvements.
Scenario 3: Limited Impact (Pessimistic)
Chai's tools help in some cases but don't generalize well across disease areas and molecule types. Early partnerships disappoint. The technology turns out to be overhyped. The company struggles to grow revenue, raises additional funding at down rounds, and eventually pivots or shuts down. Some of its IP is valuable, and might be acquired by larger companies, but the standalone company doesn't become a major player.
Based on the evidence (strong team, major pharma partnerships, investor confidence, enabling technology maturity), Scenario 2 seems most likely. Scenario 1 is possible but would require everything to go very well. Scenario 3 is possible if the technology disappoints in practice, but current evidence suggests otherwise.

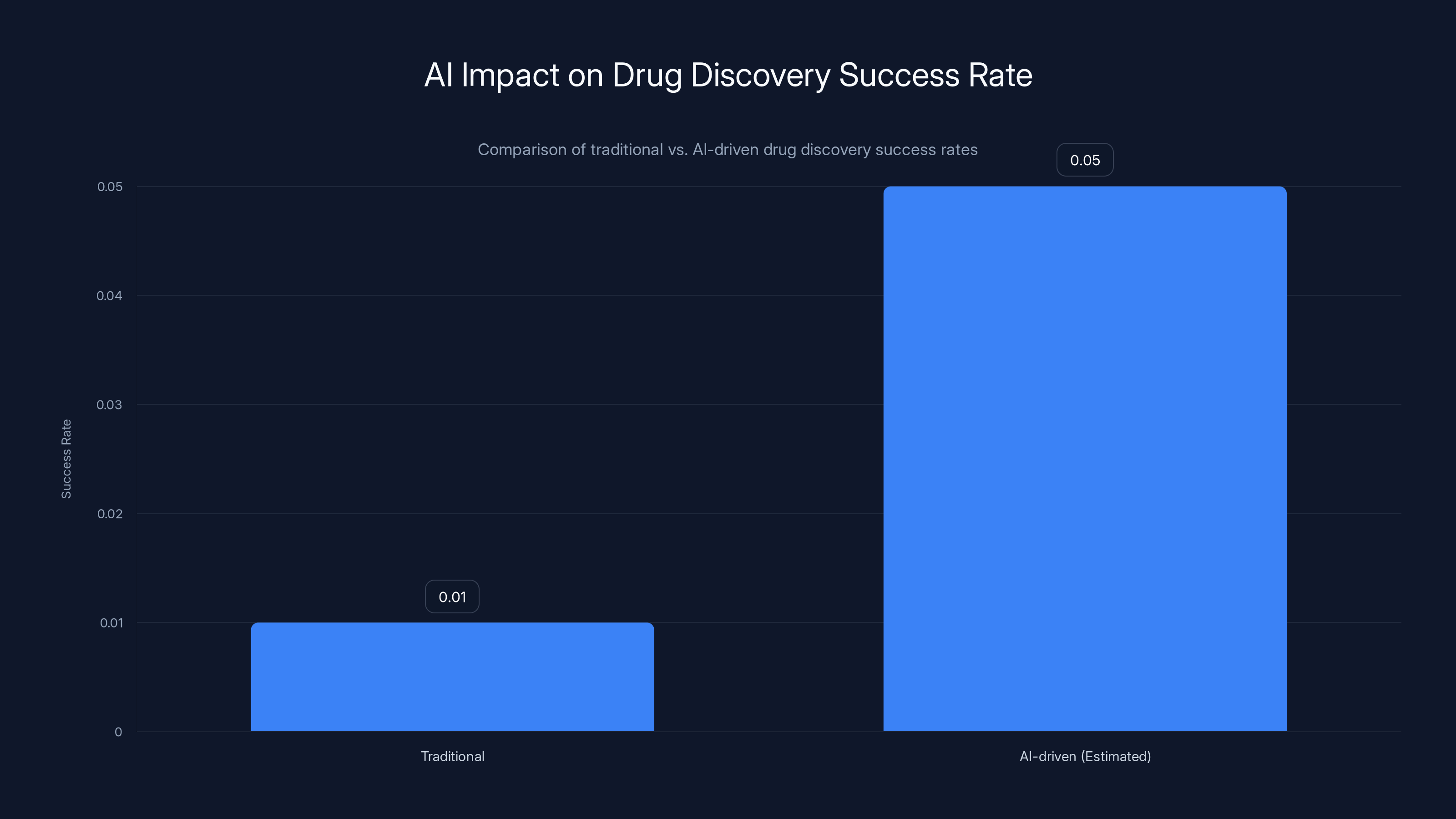
AI-driven drug discovery is estimated to increase the success rate from 0.01% to 0.05%, potentially revolutionizing the pharmaceutical industry. Estimated data.
Implications for the Future of Medicine
Beyond Chai's specific story, what does the rise of AI-powered drug discovery mean for the future of medicine?
One implication is that drug discovery will become more efficient. The cost and time to develop drugs will decrease, though probably not disappear. This means more diseases will become economically viable to treat. We'll see more drugs for rare diseases. We'll see more personalized medicines—drugs tailored to individual genetic profiles. We'll see faster innovation, with new classes of drugs emerging more frequently.
Another implication is that the structure of the pharmaceutical industry might change. Traditionally, large pharma companies have large research departments that do drug discovery in-house. If AI tools democratize discovery—if a smaller company can discover drugs as efficiently as a large company—we might see more distributed drug development. Smaller biotech companies might be able to compete with large pharma on discovery. Or large pharma might increasingly outsource discovery to specialized AI companies, focusing their internal resources on development and commercialization.
A third implication is that the timeline for pandemic response might improve. When a new pathogen emerges (like COVID-19), drug discovery is a bottleneck. If AI tools could compress discovery timelines from years to months, public health responses could be faster. This has significant geopolitical implications.
A fourth implication is that the nature of scientific discovery might change. Historically, drug discovery has been an art as much as a science. Medicinal chemists would make intuitive leaps, try unconventional approaches, and stumble onto breakthroughs. As AI becomes more involved in the process, discovery becomes more systematized, more computational, more data-driven. This is good for efficiency but might affect the types of breakthroughs that happen. Highly non-intuitive, unexpected discoveries might be less likely if the process is more algorithmic.
Competitive Dynamics and the Broader Ecosystem
Chai isn't alone in the AI drug discovery space, and understanding the competitive landscape is important for assessing the company's long-term prospects.
Some competitors are specialized by molecule type. Some focus on small molecules (traditional chemical drugs), others on biologics (protein-based drugs) like Chai. Some are specialized by disease area. Some are building foundational tools (like protein structure prediction), while others are building applied tools for specific discovery phases.
Some of the competitive dynamics:
-
Large AI companies entering the space: Google DeepMind, Microsoft Research, Meta, and other large AI companies have biotech research groups. They could apply their AI expertise to drug discovery. However, these companies might focus on research or partnership rather than building consumer-facing drug discovery platforms.
-
Biotech startups building their own AI: Traditional biotech companies are also hiring AI talent and building internal capabilities. Rather than outsourcing to companies like Chai, they might build AI tools tailored to their specific pipeline.
-
Pharma companies partnering widely: As we see with Eli Lilly (partnering with both Chai and Nvidia), major pharma companies are likely to partner with multiple AI companies and research groups to ensure they have access to the best tools.
-
Open science contributions: Academic researchers are publishing breakthroughs in protein language models, generative models for biology, and related areas. These open science contributions raise the tide for all boats—they improve the overall capability of the ecosystem. Chai's proprietary innovations are valuable, but so are open science contributions that the entire ecosystem can build on.

Why Startups Win (and Sometimes Lose) in Emerging Technology Markets
Chai's success so far reflects some well-documented patterns about startups in emerging technology markets.
Startups often win because they're organized around new technology from day one. Established companies have legacy processes, legacy infrastructure, and legacy thinking. They're optimized for their current business model. When a new technology emerges, it often threatens that business model, so large companies move slowly to embrace it. Startups can move fast because they have no legacy burden.
Chai was founded when transformer models, protein language models, and generative AI had all reached maturity. The founders built the company around this new technology from day one. If Eli Lilly had tried to build equivalent technology internally, it would have had to retrain people, reorganize teams, and overcome internal politics. By partnering with Chai, Eli Lilly gets a team that's entirely focused on the new technology.
Startups also win by recruiting top talent. When an exciting new company is founded by impressive founders working on an important problem, top researchers and engineers want to join. Chai recruited talent from OpenAI, Facebook, and other leading institutions. These people chose Chai over staying at established companies because they believed in the mission and the potential.
Startups can also move faster on decision-making. Pharma companies have complex internal review processes, IP ownership issues, and organizational politics. Startups can make decisions faster. This matters in emerging fields where the tech landscape is changing quickly.
That said, startups also have disadvantages. They lack the resources, distribution, and credibility that established companies have. This is why partnerships are so important. By partnering with Eli Lilly, Chai gains credibility, gets access to resources and data, and gets help with commercialization.
Investment Implications and Market Lessons
For investors, Chai's story offers several lessons:
1. Technology inflection points create opportunities. Chai was founded at a moment when underlying AI technology (transformers, language models) had matured enough to be practically applied to biology. Investors who identify these inflection points—moments when enabling technology crosses a threshold—can find companies at the beginning of massive value creation.
2. Team and experience matter immensely. Chai's team had worked at OpenAI (AI expertise), Facebook (protein language models), Stripe (entrepreneurship), and Absci (biotech application). This combination was hard to replicate. Investing in founders with deep expertise in both the technology and the domain is often a good bet.
3. Validation from customers is crucial. Eli Lilly's partnership is worth more than marketing claims. Real customers using your product, making business decisions based on it, and paying for it, is the gold standard of validation. VCs invest based on the strength of customer interest.
4. Large addressable market matters. Drug discovery is a multi-hundred-billion-dollar market. Even if Chai captures a small percentage of this market, the company could be worth tens of billions of dollars. VCs want to invest in large markets where the winners can be huge.
5. Multiple revenue streams are attractive. Chai could potentially generate revenue through: (a) licensing deals with pharma companies, (b) research partnerships, (c) tool subscriptions if they build SaaS products, (d) royalties on successful drugs. This flexibility reduces risk.

Risks and Challenges Ahead
Despite the impressive trajectory, Chai faces real challenges:
1. Execution risk. The Eli Lilly partnership has promised results (molecules in clinical trials by 2027). If those timelines slip or the results disappoint, confidence will erode.
2. Technology risk. AI-designed molecules might not work as well in practice as the models predict. Unexpected toxicity, manufacturing issues, or biological effects could emerge.
3. Competition. Other startups and established companies will try to build competing technology. Some might succeed. The space is unlikely to have a single winner.
4. Regulatory risk. Regulators might require additional scrutiny of AI-designed drugs, or might move slowly on approving them. This would delay the commercialization timeline.
5. Data availability. As discussed, Chai's models will improve with more data. But if pharma companies limit data sharing or don't provide high-quality data, model improvement will be slow.
6. IP and patent risk. Patent disputes over AI-designed drug patents could be complex. Who owns the IP when a drug is designed using AI? Questions like this will likely end up in court.
The Broader Narrative: AI Transforming Industries
Chai's story is one example of a broader narrative: artificial intelligence is transforming industry after industry. We've seen it in images (computer vision), text (language models), games (AlphaGo, AlphaZero), and now biology and drug discovery.
The pattern is consistent: an AI capability matures (usually in a research lab or at a large tech company), a startup is founded to commercialize it, the startup raises capital and recruits talent, partnerships form with established companies, and value creation begins.
This pattern doesn't always work out—many startups fail, and hype often outpaces reality. But when the technology is real and the application is valuable, the results can be transformative.
Chai is betting that AI can transform drug discovery, just as transformers transformed language understanding, and AlphaFold transformed protein structure prediction. If they're right, the company will be enormously valuable and will contribute to a significant improvement in human health. If they're wrong, the company will fail and investors will learn lessons for the next venture.
The fact that major pharma companies like Eli Lilly are willing to bet billions suggests the smart money thinks Chai is onto something real.

FAQ
What exactly does Chai-2, the algorithm, do?
Chai-2 is an AI model trained to generate antibody sequences (the proteins that fight disease) that are likely to bind effectively to disease targets. Rather than requiring scientists to screen millions of compounds experimentally, the algorithm generates high-quality candidates computationally. Think of it as a designer tool for molecular structures, where instead of manually trying thousands of combinations, the AI predicts which combinations will work best before anything is synthesized.
How did Chai Discovery get founded, and who are the founders?
Chai was founded in 2024 by Josh Meier, Jack Dent, Matthew Mc Partlon, and Jacques Boitreaud. The story began six years earlier when Sam Altman from OpenAI approached Meier and Dent about starting a proteomics company. The timing wasn't right then, but by 2024—after Meier had worked at Facebook on protein language models and at Absci on biotech—the founders felt the AI technology had matured enough. OpenAI became an early investor, and the founders worked out of OpenAI's San Francisco offices initially.
What's the relationship between Chai Discovery and Eli Lilly?
Eli Lilly announced a strategic partnership with Chai where the pharmaceutical company will use Chai's generative design models combined with Lilly's internal biologics expertise and proprietary data. The goal is to design better antibodies faster and get molecules into clinical trials more quickly. This partnership validates Chai's technology at the highest level—one of the world's largest pharma companies is betting its drug pipeline on the startup's tools.
Could AI drug discovery actually compress development timelines from 10 years to a few years?
Fully compressing drug development from 10-15 years to just a few years is unlikely. However, AI tools can meaningfully accelerate the discovery phase (identifying a promising molecule) and early optimization phase, potentially saving 1-3 years. Clinical trials and regulatory approval still take years because they depend on collecting real-world safety and efficacy data from patients. Realistic expectations are 20-50% timeline compression, not 80-90%.
Why is this moment special for AI-powered drug discovery, compared to previous attempts?
Previous attempts to apply AI to drug discovery lacked one or more crucial ingredients: mature AI models, abundant computational resources, large training datasets, or deep understanding of protein biology. By 2024, all of these pieces had converged. Transformer models had proven revolutionary in language and now biology. Computational infrastructure was cheap and abundant. Public protein databases were massive. The moment of inflection had arrived.
What are the biggest risks that could derail Chai Discovery?
The main risks are: (1) execution risk—if the Eli Lilly partnership disappoints and doesn't produce promised results, confidence erodes; (2) technology risk—AI-designed molecules might have unforeseen toxicity or manufacturing issues; (3) competition—other startups and established companies will build competing tools; (4) regulatory risk—if regulators become skeptical of AI-designed drugs and require extra scrutiny, timelines extend; and (5) data limitation—if pharma companies don't share quality proprietary data, the models won't improve as fast as expected.
How is Chai positioned differently from other AI drug discovery companies?
Chai has several differentiators: its founding team has expertise in both cutting-edge AI (OpenAI, Facebook) and practical biotech (Absci); the company specializes in antibodies and biologics, which is a high-growth area in pharma; and most importantly, the company has secured partnership validation from Eli Lilly, one of the world's largest pharma companies. This partnership is worth more than any marketing claim—it shows an established player trusts the technology enough to build it into their pipeline.
What does the $1.3 billion valuation mean, and is it justified?
Chai raised
Could pharmaceutical companies just build equivalent AI tools internally instead of partnering with Chai?
Some pharma companies will try to build internal capabilities. However, startups typically move faster on new technology because they have no legacy systems or organizational politics to overcome. Recruiting top AI talent is also easier for a startup focused entirely on one problem than for a large company with many competing priorities. Partnership allows pharma companies to benefit from Chai's focus and speed while contributing their own expertise and data. Both benefit more than either would alone.
How will this technology affect drug costs and drug access?
In theory, if drug discovery becomes cheaper and faster, drug costs could decrease because development costs are lower. However, pharmaceutical pricing is complex and driven by many factors beyond development cost: market demand, competition, regulatory approval, manufacturing, and marketing. While AI-driven discovery might contribute to cost reduction over time, it's not a guarantee. The more likely impact is that we'll see more drugs developed for rare diseases and underserved populations, because economic viability improves when development costs decrease.
Conclusion: The Inflection Point in Drug Discovery
Chai Discovery's rapid rise from concept to billion-dollar valuation and major pharma partnership represents something significant: the convergence of artificial intelligence capability and biological application has reached a critical threshold. This isn't just another startup using AI to solve a problem. It's a moment where the fundamental economics and timelines of drug discovery might actually change.
The founding story—Sam Altman's suggestion six years ago, the founders waiting for the technology to mature, finally making the leap in 2024—illustrates an important principle about emerging technologies. Timing matters immensely. An idea that's too early fails because the technology isn't ready. An idea that's too late fails because the market is saturated. Chai appears to have arrived at the right moment.
What makes this moment special is the confluence of multiple factors. Transformer models, which revolutionized language understanding, are proving equally powerful for understanding biological sequences. Protein language models have been developed and refined over years of research. Large computational infrastructure is available and affordable. Training data about proteins and drug molecules is abundant. The AI talent pool interested in biology has grown. Major pharmaceutical companies have concluded that AI is important enough to warrant substantial investment and partnership.
The Eli Lilly partnership is particularly telling. Lilly isn't a venture-backed startup taking a bet on a long shot. It's a 148-year-old company with global reach, thousands of researchers, and billions in revenue. Lilly doesn't partner with companies unless the company's technology passes rigorous internal scrutiny. Lilly's decision to partner with Chai, and to invest $1 billion in an AI drug discovery lab with Nvidia, signals that the company's leadership believes AI will materially accelerate drug discovery.
Chai's investors are also making a specific claim: companies that move quickly to adopt AI-powered drug discovery will gain competitive advantages—they'll get molecules into clinical trials faster, and that speed will translate to competitive advantage in bringing medicines to market. This is plausible. In competitive industries, being first often matters. The pharmaceutical industry is intensely competitive. If Eli Lilly can develop certain drugs 1-2 years faster than competitors using traditional methods, that's worth billions in competitive advantage.
That said, it's important to be realistic about what this technology can and can't do. Drug discovery isn't the only bottleneck in pharmaceutical development. Clinical trials, regulatory approval, and manufacturing also take years. AI can accelerate discovery and optimization, but it can't eliminate these other phases. Timelines are likely to compress by 20-50%, not by 80-90%. This is still significant—it's worth billions—but it's not a panacea.
There are also legitimate reasons to be skeptical. Every technology area has hype cycles. AI has been oversold before. Some of Chai's claims might not pan out. The molecules it designs might not work as well in practice as the models predict. Competition might emerge. Regulatory scrutiny might slow adoption. These are all real risks.
But the weight of the evidence suggests Chai is onto something real. The team is exceptional. The partnership is real and comes from an established player with skin in the game. The underlying AI technology is proven. The market opportunity is enormous. The timing is right.
For patients, the potential upside is significant. If drug discovery becomes faster and more efficient, we'll see more medicines for diseases that are currently hard to treat. We'll see personalized medicines tailored to individual genetic profiles. We'll see innovation accelerate. Diseases that today seem intractable might yield to new approaches. That matters.
For investors, Chai represents a bet on the AI-biotech convergence. If that convergence delivers value—if AI-designed drugs make it into clinical trials and succeed—early investors in companies like Chai could see enormous returns. The valuation is high, but the upside is potentially much higher if the company executes.
For the pharmaceutical industry, Chai and companies like it represent a shift from traditional R&D to AI-augmented R&D. Over the next decade, the companies that successfully integrate AI tools into their discovery pipelines will likely outcompete those that stick with traditional methods. This will put pressure on all pharma companies to invest in AI capabilities, either internally or through partnerships. It will also change the structure of the industry—smaller, more specialized biotech companies might be able to compete with large pharma if they have good AI tools.
Chai's story is still early. The company is only 14 months old. The real test will come when Eli Lilly's partnership produces results. Do the molecules designed with Chai's tools work better? Do they move faster through development? Do they eventually become medicines that help patients? The answers to these questions will determine whether Chai was ahead of the curve or another AI startup that overpromised and underdelivered.
But based on the evidence available today—the team, the partnerships, the valuation, the investor confidence—Chai appears to be at the beginning of something significant. We're watching an inflection point in pharmaceutical R&D unfold in real time. The next few years will be fascinating to watch.
The lesson for entrepreneurs and investors is that transformative technologies often follow a pattern: enabling technology matures, founders recognize the moment, capital flows, partnerships form, and value creation begins. Chai is executing this playbook skillfully. Whether the execution continues and the promise is realized will be determined in the coming years, but the groundwork appears solid.

Key Takeaways
- Chai Discovery achieved unicorn status ($1.3B valuation) in just 14 months through exceptional team, enabling technology maturity, and Eli Lilly partnership validation
- The founders waited 6 years for AI technology (transformers, protein language models) to mature enough before founding the company, demonstrating patience and strategic timing
- Chai-2 algorithm generates antibody candidates computationally using generative AI, compressing discovery timelines by 20-50% compared to traditional high-throughput screening
- Eli Lilly's partnership signals major pharmaceutical companies are moving from AI skepticism to integration, expecting first-in-class medicines in clinical trials by 2027
- The market opportunity is enormous—drug discovery is a multi-hundred-billion-dollar industry, and timeline/cost compression could unlock treatment for rare diseases and accelerate innovation
![How Chai Discovery Became AI Drug Development's Rising Star [2025]](https://tryrunable.com/blog/how-chai-discovery-became-ai-drug-development-s-rising-star-/image-1-1768595981206.jpg)



